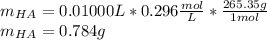Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 04:00
14. many depressants reduce small muscle control, making it harder for a. you to steer b. your mind to consider complex problems c. the eye to scan, focus, or stay still d. the kidneys to filter alcohol out of the bloodstream
Answers: 3

Chemistry, 22.06.2019 04:50
The name of the ion, s2-, is: sulfurous ion sulfide ion sulfur ion sulfate ion
Answers: 1

Chemistry, 22.06.2019 05:40
Salicylic acid is a very important acid. it is used to synthesize the aspirin by treating with acetic anhydride. a 0.2015-g sample of salicylic acid was dissolved in a 100.00-ml volumetric flask, and the solution was diluted to the mark. a 10-ml aliquot of this solution was titrated with standard naoh (0.01130 + 0.2% n) to a phenolphthalein faint pink color end point at 19.81 ml. (a) (calculate the normality of the salicylic acid solution used in the titration. (b) assuming the salicylic acid is pure, what is the equivalent weight of the salicylic acid? practice problems for the final exam (continued) (c) (calculate the inherent error in the determination of the equivalent weight you calculated in part (b). use the following absolute errors in the equipment /glassware when calculating the inherent error. 5.00-ml pipet: + 0.02 ml 100-ml volumetric flask: + 0.08 ml analytical balance: + 0.2 mg 25-ml buret: + 0.03 ml
Answers: 2

Chemistry, 22.06.2019 09:20
Sugar is dissolved in water. which is the solute? sugar neither both water
Answers: 1
You know the right answer?
Thiamine hydrochloride (C12H18ON4SCl2) is a water-soluble form of thiamine (vitamin B1; Ka = 3.37×10...
Questions

Social Studies, 16.09.2019 01:30

Mathematics, 16.09.2019 01:30

Mathematics, 16.09.2019 01:30

History, 16.09.2019 01:30


Arts, 16.09.2019 01:30

History, 16.09.2019 01:30



Mathematics, 16.09.2019 01:30

Health, 16.09.2019 01:30

Mathematics, 16.09.2019 01:30


Biology, 16.09.2019 01:30

Biology, 16.09.2019 01:30


English, 16.09.2019 01:30



History, 16.09.2019 01:30

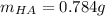
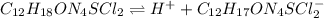
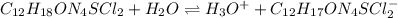
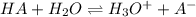
![pH=pKa+log(\frac{[A^-]}{[HA]} )](/tpl/images/0553/3571/4a01a.png)
![log(\frac{[A^-]}{[HA]} )=3.50-[-log(3.37x 10^{-7})]=3.50-6.47=-2.97}\\\\\frac{[A^-]}{[HA]} =10^{-2.97}=1.07x10^{-3}](/tpl/images/0553/3571/b4cdb.png)
![[A^-]=1.07x10^{-3}}[HA]](/tpl/images/0553/3571/1acb8.png)
![[H]^+=[A^-]=10^{-pH}=10^{-3.50}=3.16x10^{-4}M](/tpl/images/0553/3571/d3a0e.png)
![[HA]=\frac{3.16x10^{-4}M}{1.07x10^{-3}} =0.296M](/tpl/images/0553/3571/7981c.png)