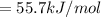Chemistry, 19.03.2020 05:55 bryanmcmillianjr
Consider the Gibbs energies at 25 ∘ C. Substance Δ G ∘ f ( kJ ⋅ mol − 1 ) Ag + ( aq ) 77.1 Cl − ( aq ) − 131.2 AgCl ( s ) − 109.8 Br − ( aq ) − 104.0 AgBr ( s ) − 96.9 (a) Calculate Δ G ∘ rxn for the dissolution of AgCl ( s ) .

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 04:00
Mitosis is a type of cell division that produces cells that are identical to the parent cell. meiosis is a different type of cell division that produces cells that carry have a genetic material of the parent cell. based on the information provided how do the purpose of mitosis and meiosis differ
Answers: 3

Chemistry, 22.06.2019 09:20
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1

Chemistry, 22.06.2019 13:00
Using the thermodynamic information in the aleks data tab, calculate the standard reaction free energy of the following chemical reaction: →+p4o10s6h2ol4h3po4s round your answer to zero decimal places.
Answers: 3
You know the right answer?
Consider the Gibbs energies at 25 ∘ C. Substance Δ G ∘ f ( kJ ⋅ mol − 1 ) Ag + ( aq ) 77.1 Cl − ( aq...
Questions

Mathematics, 31.08.2019 17:10

Mathematics, 31.08.2019 17:10



Mathematics, 31.08.2019 17:10

Social Studies, 31.08.2019 17:10

Biology, 31.08.2019 17:10

Social Studies, 31.08.2019 17:10


Social Studies, 31.08.2019 17:10



Geography, 31.08.2019 17:10


Business, 31.08.2019 17:10

Biology, 31.08.2019 17:10




![\Delta G_{rxn}^o=\sum[\Delta G^o_{f}]_{products}-\sum[\Delta G^o_{f}]_{reactants}](/tpl/images/0553/6074/2b279.png)
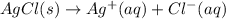
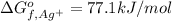
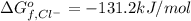
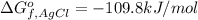
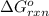 :
:![\Delta G_{rxn}^o=[77.1 kJ/mol+(-131.2 kJ/mol)]-[-109.8 kJ/mol]](/tpl/images/0553/6074/68588.png)