
A piece of iron (mass = 25.0 g) at 398 K is placed in a styrofoam coffee cup containing 25.0 mL of water at 298 K. Assuming that no heat is lost to the cup or the surroundings, what will the final temperature of the water be? The specific heat capacity of iron = 0.449 J/g°C and water = 4.18 J/g°C.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Which traits do human embryos have that link them to a common ancestor with fish and reptiles? a. scales and tail b. gill slits and scales c. tail and gill slits d. hair and tail
Answers: 2

Chemistry, 21.06.2019 22:30
Which statement best describes the oxidation numbers of the atoms found in magnesium chloride? a. magnesium has a 2- oxidation number and chlorine has a 1+ oxidation number. b. magnesium has a 2- oxidation number and chlorine has a 2+ oxidation number. c. magnesium has a 2+ oxidation number and chlorine has a 1- oxidation number. d. magnesium has a 1+ oxidation number and chlorine has a 1- oxidation number.
Answers: 2

Chemistry, 22.06.2019 05:00
Frictional forces acting on an object are often converted into energy, which causes the temperature of the object to rise slightly.
Answers: 2

You know the right answer?
A piece of iron (mass = 25.0 g) at 398 K is placed in a styrofoam coffee cup containing 25.0 mL of w...
Questions

Physics, 16.10.2019 03:00


Engineering, 16.10.2019 03:00


Biology, 16.10.2019 03:00

French, 16.10.2019 03:00

Mathematics, 16.10.2019 03:00


Geography, 16.10.2019 03:00


Mathematics, 16.10.2019 03:00


Mathematics, 16.10.2019 03:00

Mathematics, 16.10.2019 03:00

Mathematics, 16.10.2019 03:00




Mathematics, 16.10.2019 03:00

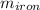 = 25 gm
= 25 gm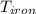 = 398 K
= 398 K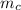 = 25 ml = 25 gm
= 25 ml = 25 gm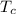 = 298 K
= 298 K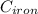 = 0.449
= 0.449 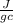
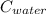 = 4.18
= 4.18 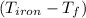 =
= 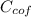 ×
× 
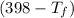 = 25 × 4.18 ×
= 25 × 4.18 × 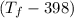
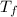 = 9.3 (
= 9.3 ( 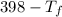 = 9.3
= 9.3 


