Chemistry, 19.03.2020 17:08 Levantine3667
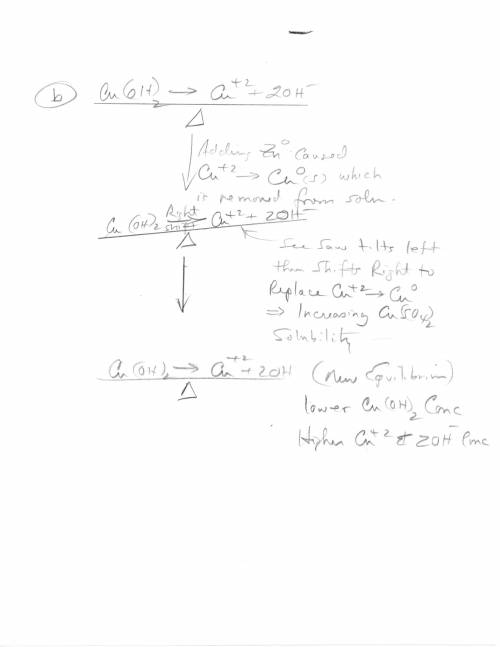
3. Copper hydroxide dissolves in water according to the following equation. It is only very slightly soluble. Cu(OH)2(s) Cu2 (aq) 2OH- (aq) a. Explain how the solubility can be increased by adding HCl to the solution. b. Explain how the concentration of copper (II) ion or of hydroxide ion can be reduced in the solution so that more of the solid copper hydroxide can be dissolved.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 13:10
Which of these is the result of scientific research and not engineering? a. a new shoe design that features air cushioning for more comfort and protection b. the creation of glass with uv protection. c. a conclusion about diet commonalities among diabetics. d. the development of a smaller, more compact missile.
Answers: 1

Chemistry, 22.06.2019 14:30
In water, a strong acid will break down into its component parts. a. completely b. partly c. never in water, a weak base will break down into its component parts. a. completely b. partly c. never
Answers: 2


Chemistry, 22.06.2019 15:30
Which statement names the physical property of wood a. wood is softer than coal b. wood does not rust c. wood can rot d. wood can burn
Answers: 1
You know the right answer?
3. Copper hydroxide dissolves in water according to the following equation. It is only very slightly...
Questions


Mathematics, 24.02.2021 06:10


Chemistry, 24.02.2021 06:10



Mathematics, 24.02.2021 06:10




Mathematics, 24.02.2021 06:10


History, 24.02.2021 06:10



Mathematics, 24.02.2021 06:10

Mathematics, 24.02.2021 06:10

Mathematics, 24.02.2021 06:10


Mathematics, 24.02.2021 06:10