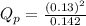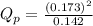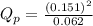Chemistry, 19.03.2020 22:42 Nyasiabaltimore3
He equilibrium constant (KP) is 0.21 at a particular temperature for the reaction:
N2O4(g) <=> 2NO2(g)
Given the following sets of initial conditions, what is the net change that must occur for the reaction to reach equilibrium? Does the reaction shift left to reach equilibrium, does the reaction shift right to reach equilibrium or is the reaction at equilibrium at these initial concentrations so no net change will occur?
Options: Equilibrium, Left, or Right
a. PNO2 = 0.225 atm, PN2O4 = 0.134 atm
b. PNO2 = 0.086 atm, PN2O4 = 0.064 atm
c. PNO2 = 0.130 atm, PN2O4 = 0.142 atm
d. PNO2 = 0.173 atm, PN2O4 = 0.142 atm
e. PNO2 = 0.151 atm, PN2O4 = 0.062 atm

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:30
You have a sample of a gas that occupies a volume of 17ml at -111 degrees celsius. what volume does the sample occupy at 88 degrees celsius? show all work asap
Answers: 3


Chemistry, 22.06.2019 06:00
Ethanol (c2h5oh) is produced from the fermentation of sucrose in the presence of enzymes. c12h22o11(aq) + h2o(g) 4 c2h5oh(l) + 4 co2(g) determine the theoretical yield and the percent yields of ethanol if 680. g sucrose undergoes fermentation and 326.5 g ethanol is obtained. theoretical _ g _ percent %
Answers: 1

Chemistry, 22.06.2019 16:00
Click the button that shows the correct relationship of the electron affinities of the elements sodium and phosphorus. sodium’s electron affinity value is more negative than the electron affinity value of phosphorus. phosphorus’ electron affinity value is more negative than the electron affinity value of sodium. this information cannot be determined using the periodic table. answer is b on e2020.
Answers: 3
You know the right answer?
He equilibrium constant (KP) is 0.21 at a particular temperature for the reaction:
N2O4...
N2O4...
Questions


German, 12.04.2020 02:39


Biology, 12.04.2020 02:39

Mathematics, 12.04.2020 02:39


Mathematics, 12.04.2020 02:39

History, 12.04.2020 02:39

Mathematics, 12.04.2020 02:39

English, 12.04.2020 02:39


English, 12.04.2020 02:40


History, 12.04.2020 02:40

Spanish, 12.04.2020 02:40



Mathematics, 12.04.2020 02:40

Mathematics, 12.04.2020 02:40

English, 12.04.2020 02:40


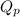 as follows.
as follows.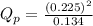
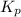 is 0.21 (which is smaller than the value) this means that equilibrium will shift to the left
.
is 0.21 (which is smaller than the value) this means that equilibrium will shift to the left
.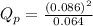
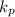 which means that the equilibrium will shift to the right.
which means that the equilibrium will shift to the right.