
For the reaction CO2(g) + H2O(ℓ) + CaCl2(aq) ←→ CaCO3(s) + HCl(aq) K = 4.8 the concentrations of CO2, H2O, CaCl2 and HCl are 0.377 M, 55.4 M, 0.652 M and 30.2 M, respectively. What will happen in order for the system to reach equilibrium? 1. The reaction will shift to the right. 2. Nothing will occur. 3. The reaction will shift to the left. 4. Not enough information is given.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 01:30
Reaction rate depends on how many molecules are coming into contact with each other with enough energy to react. increasing the temperature of the reactants will increase -
Answers: 3

Chemistry, 22.06.2019 07:30
What three things determine the shape and size of a puddle when water is poured out onto a surface
Answers: 2


Chemistry, 23.06.2019 07:50
What is the significance sodium hydroxide and hydrochloric acid
Answers: 1
You know the right answer?
For the reaction CO2(g) + H2O(ℓ) + CaCl2(aq) ←→ CaCO3(s) + HCl(aq) K = 4.8 the concentrations of CO2...
Questions

Mathematics, 11.10.2020 02:01

Mathematics, 11.10.2020 02:01

Mathematics, 11.10.2020 02:01

Biology, 11.10.2020 02:01

Mathematics, 11.10.2020 02:01

Mathematics, 11.10.2020 02:01

Mathematics, 11.10.2020 02:01



Mathematics, 11.10.2020 02:01

Mathematics, 11.10.2020 02:01





Mathematics, 11.10.2020 02:01




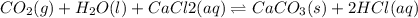
![K=\frac{[HCl]^2}{[CaCl_2]}](/tpl/images/0554/8954/74ccf.png)
![Q=\frac{[HCl]^2}{[CaCl_2]}=\frac{ 30.2^2 }{0.652 }=1398.8](/tpl/images/0554/8954/f4b79.png)


