
Chemistry, 20.03.2020 00:06 buddyclayjohnson
A mixture of methane (CH4) and ethane (C2H6) is stored in a container at 294 mm Hg. The gases are burned in air to form CO2 and H2O. If the pressure of CO2 is 351 mm Hg measured at the same temperature and volume as the original mixture, calculate the mole fraction of the gases.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:10
What does a particular point on a line of a phase diagram represent? o a. the maximum temperature a substance can exist at without bonds breaking b. the pressure created by the kinetic energy of molecules at a particular temperature c. the melting point or boiling point of a substance at a specific pressure d. the conditions in which temperature and pressure have equal effects on a substance
Answers: 2


Chemistry, 22.06.2019 16:30
Ammonium perchlorate nh4clo4 is the solid rocket fuel used by the u.s. space shuttle. it reacts with itself to produce nitrogen gas n2 , chlorine gas cl2 , oxygen gas o2 , water h2o , and a great deal of energy. what mass of nitrogen gas is produced by the reaction of 2.1g of ammonium perchlorate?
Answers: 2

You know the right answer?
A mixture of methane (CH4) and ethane (C2H6) is stored in a container at 294 mm Hg. The gases are bu...
Questions






Physics, 01.12.2020 01:00


Mathematics, 01.12.2020 01:00



Chemistry, 01.12.2020 01:00

Mathematics, 01.12.2020 01:00



English, 01.12.2020 01:00

Mathematics, 01.12.2020 01:00




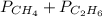 = 294 mm Hg
= 294 mm Hg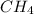 will yield 1 mole of
will yield 1 mole of 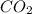 whereas 1 mole of
whereas 1 mole of 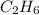 will yield 2 moles of
will yield 2 moles of 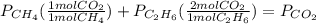
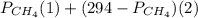 = 351 mm Hg
= 351 mm Hg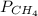 = 2(294) - 351
= 2(294) - 351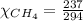
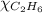 = 1 - 0.806
= 1 - 0.806

