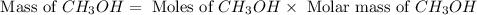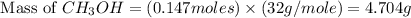Based on the ideal gas law, there is a simple equivalency that exists between the amount of gas and the volume it occupies. At standard temperature and pressure (STP; 273.15 K and 1 atm, respectively), one mole of gas occupies 22.4 L of volume. What mass of methanol (CH3OH) could you form if you reacted 6.59 L of a gas mixture (at STP) that contains an equal number of carbon monoxide (CO) and hydrogen gas (H2) molecules?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 22:30
Essay-alternative energy sources research sources of energy that are being developed. write a report of 350-400 words discussing the information you learned concerning the development of various energy sources and the impact that you think they will have on your life. include sources cited at the end of your report using the mla format. follow the rubric guidelines. note that wikipedia is not an appropriate resource for a research paper. worth 99
Answers: 3


Chemistry, 23.06.2019 04:30
Two liquids are poured into a beaker. after a few seconds, the beaker becomes warm. which of the following best describes this reaction? a. an exothermic reaction b. a decomposition reaction c. an endothermic reaction d. a single-displacement reaction
Answers: 1

Chemistry, 23.06.2019 09:00
The vapor pressure of water at 25.0°c is 23.8 torr. determine the mass of glucose (molar mass = 180 g/mol) needed to add to 500.0 g of water to change the vapor pressure to 22.8 torr.
Answers: 1
You know the right answer?
Based on the ideal gas law, there is a simple equivalency that exists between the amount of gas and...
Questions





Mathematics, 22.01.2021 03:40

Health, 22.01.2021 03:40

Mathematics, 22.01.2021 03:40


Mathematics, 22.01.2021 03:40

Mathematics, 22.01.2021 03:40

Mathematics, 22.01.2021 03:40

Mathematics, 22.01.2021 03:40

Mathematics, 22.01.2021 03:40


Mathematics, 22.01.2021 03:40

Biology, 22.01.2021 03:40

Mathematics, 22.01.2021 03:40


Mathematics, 22.01.2021 03:40

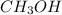 produced is, 4.704 grams.
produced is, 4.704 grams.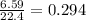 mole of gas mixture
mole of gas mixture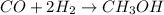
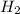 react with 1 mole of
react with 1 mole of 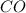
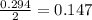 moles of
moles of