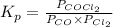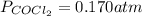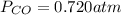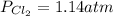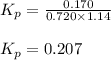Chemistry, 20.03.2020 01:39 wwwserban9281
Phosgene (carbonyl chloride), COCl2, is an extremely toxic gas that is used in manufacturing certain dyes and plastics. Phosgene can be produced by reacting carbon monoxide and chlorine gas at high temperatures: CO(g)+Cl2(g)⇌COCl2(g) Carbon monoxide and chlorine gas are allowed to react in a sealed vessel at 468 ∘C . At equilibrium, the concentrations were measured and the following results obtained: Gas Partial Pressure (atm) CO 0.720 Cl2 1.14 COCl2 0.170 What is the equilibrium constant, Kp, of this reaction?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 13:40
Which are causes of mechanical weathering? check all that apply.oacid raino plant growtho animal actionso carbon dioxideo pressure release
Answers: 1

Chemistry, 22.06.2019 23:00
What is the mass of naoh that would have to be added to 500 ml of a solution of 0.20 m acetic acid in order to achieve a ph of 5.0?
Answers: 1

Chemistry, 23.06.2019 07:00
Introduction of drugs into the gastrointestinal tract is a form of administration. a. enteral b. topical c. parenteral d. inhalation
Answers: 1

Chemistry, 23.06.2019 10:30
Me soon im confused much mass would a mole of hydrogen molecules contain? recall that hydrogen is diatomic. g/mol
Answers: 1
You know the right answer?
Phosgene (carbonyl chloride), COCl2, is an extremely toxic gas that is used in manufacturing certain...
Questions




Mathematics, 29.07.2019 05:40

Chemistry, 29.07.2019 05:40

History, 29.07.2019 05:40

History, 29.07.2019 05:40

Arts, 29.07.2019 05:40

Arts, 29.07.2019 05:40

Arts, 29.07.2019 05:40






Mathematics, 29.07.2019 05:40




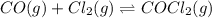
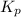 for above reaction follows:
for above reaction follows: