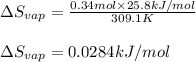Chemistry, 20.03.2020 07:24 ramanpreet
The molar heat of vaporization of pentane is 25.8 kJ·mol−1, and the boiling point of pentane is 36.1°C. Calculate the value of ΔvapS for the vaporization of 0.34 mole of pentane.

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 03:30
Adrop of acetone (nail polish remover) has a mass of 35 mg and a density of 0.788 g/cm3. what is its volume in cubic centimeters?
Answers: 3

Chemistry, 22.06.2019 07:30
Given that 1 mi = 1760 yd, determine what conver- sion factor is appropriate to convert 1849 yd to miles; to convert 2.781 mi to yards.
Answers: 2

Chemistry, 22.06.2019 16:10
Amixture initially contains a, b, and c in the following concentrations: [a] = 0.300 m , [b] = 1.05 m , and [c] = 0.550 m . the following reaction occurs and equilibrium is established: a+2b⇌c at equilibrium, [a] = 0.140 m and [c] = 0.710 m . calculate the value of the equilibrium constant, kc.
Answers: 1
You know the right answer?
The molar heat of vaporization of pentane is 25.8 kJ·mol−1, and the boiling point of pentane is 36.1...
Questions



English, 25.01.2020 00:31






Computers and Technology, 25.01.2020 00:31











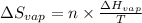
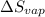 = Entropy change of vaporization = ?
= Entropy change of vaporization = ?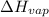 = molar heat of vaporization = 25.8 kJ/mol
= molar heat of vaporization = 25.8 kJ/mol![36.1^oC=[36.1+273]K=309.1K](/tpl/images/0555/6193/0c07c.png)