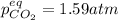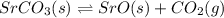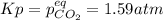Answers: 3
Another question on Chemistry



Chemistry, 22.06.2019 07:30
Calculate the ratio of h+ ions to oh– ions at a ph = 7. find the concentration of h+ ions to oh– ions listed in table b of your student guide. then divide the h+ concentration by the oh– concentration. record this calculated ratio in table a of your student guide. compare your approximated and calculated ratios of h+ ions to oh– ions at a ph = 7. are they the same? why or why not? record your comparison in table a. what is the concentration of h+ ions at a ph = 7? mol/l what is the concentration of oh– ions at a ph = 7? mol/l what is the ratio of h+ ions to oh– ions at a ph = 7? : 1
Answers: 1

Chemistry, 22.06.2019 08:30
Which of the following would have less momentum than a 52 kg cheetah running at 10 m/s?
Answers: 2
You know the right answer?
For the reaction below, Kp = 1.59 at 100°C. If 1.0 g of SrCO3 is placed in an empty 5.00 L reactor a...
Questions





English, 23.08.2019 04:20

Physics, 23.08.2019 04:20

Chemistry, 23.08.2019 04:20

Mathematics, 23.08.2019 04:20

Mathematics, 23.08.2019 04:20

Mathematics, 23.08.2019 04:20





Mathematics, 23.08.2019 04:20





Computers and Technology, 23.08.2019 04:20