Chemistry, 20.03.2020 09:07 khalilh1206
The following reaction was performed in a sealed vessel at 772 ∘C : H2(g)+I2(g)⇌2HI(g) Initially, only H2 and I2 were present at concentrations of [H2]=3.90M and [I2]=2.95M . The equilibrium concentration of I2 is 0.0500 M . What is the equilibrium constant, Kc, for the reaction at this temperature?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 19:00
Consider the point on the plot where 10.0 g of naoh have been added. what amount of naoh, in moles, has been added? 0.308 mol fecl3 initially present
Answers: 1


Chemistry, 22.06.2019 16:30
Asample of freon gas has a volume of 2.23 liters, a pressure of 4.85 kpa, and a temperature of -1.36°c. calculate the volume at a pressure of 1.38 kpa and a temperature of 5.5°c. (show work)
Answers: 1

Chemistry, 22.06.2019 18:30
Two people each hold the end of a rope and create waves by moving their arms up and down. this wave is best classified as a transverse wave because a) both the rope particles and the wave are moving in the same direction. b) the wave is moving up and down as the particles of the rope move horizontally. c) the wave is moving horizontally as the particles of the rope move up and down. eliminate d) the wave is moving in a parallel direction with the motion of the person's arms.
Answers: 3
You know the right answer?
The following reaction was performed in a sealed vessel at 772 ∘C : H2(g)+I2(g)⇌2HI(g) Initially, on...
Questions

Chemistry, 12.02.2021 17:40

Mathematics, 12.02.2021 17:40

Mathematics, 12.02.2021 17:40

Mathematics, 12.02.2021 17:40


Mathematics, 12.02.2021 17:40



Mathematics, 12.02.2021 17:40

Mathematics, 12.02.2021 17:40


Mathematics, 12.02.2021 17:40



Mathematics, 12.02.2021 17:40


Chemistry, 12.02.2021 17:40

Mathematics, 12.02.2021 17:40

Arts, 12.02.2021 17:40

Mathematics, 12.02.2021 17:40

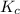 for the given equation is 160.2
for the given equation is 160.2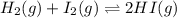
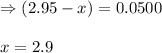
![K_c=\frac{[HI]^2}{[H_2][I_2]}](/tpl/images/0555/7227/62646.png)