
Chemistry, 20.03.2020 10:19 SMURFETTE86
For Questions 1 through 4 - Consider a solution prepared by combining 45.0 g of BaCl2 with enough water to prepare a 750.0 mL of solution. Assume that the volume of water added was 748 mL. Density of water is 1.00 g/mL. Answer questions 1 through 4. 1) What is the molarity of BaCl2 in the solution?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 04:30
Electrons are extremely important to what area of technology? a) anti-aging research b) household product development c) electronics d) drug discovery
Answers: 3

Chemistry, 22.06.2019 06:30
If 1.8 l of water is added to 2.5l of a 7.0 molarity koh solution, what is the molarity of the new solution
Answers: 1

Chemistry, 22.06.2019 12:50
What is the chemical name of the compound na2co3? use the list of polyatomic ions and the periodic table to you answer. a. sodium carbon oxide b. sodium carbonate c. sodium(ll) carbonate d. sodium oxalate
Answers: 1

Chemistry, 22.06.2019 13:50
Abeaker with 2.00×102 ml of an acetic acid buffer with a ph of 5.000 is sitting on a benchtop. the total molarity of acid and conjugate base in this buffer is 0.100 m. a student adds 4.70 ml of a 0.360 m hcl solution to the beaker. how much will the ph change? the pka of acetic acid is 4.740.
Answers: 1
You know the right answer?
For Questions 1 through 4 - Consider a solution prepared by combining 45.0 g of BaCl2 with enough wa...
Questions

Mathematics, 12.04.2021 16:00



French, 12.04.2021 16:00





Health, 12.04.2021 16:00

Mathematics, 12.04.2021 16:00

Mathematics, 12.04.2021 16:00

Geography, 12.04.2021 16:00

History, 12.04.2021 16:00

Mathematics, 12.04.2021 16:00






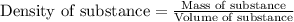
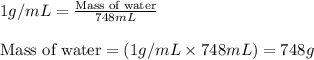
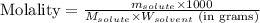
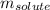 = Given mass of solute (barium chloride) = 45.0 g
= Given mass of solute (barium chloride) = 45.0 g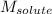 = Molar mass of solute (barium chloride) = 208.23 g/mol
= Molar mass of solute (barium chloride) = 208.23 g/mol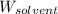 = Mass of solvent (water) = 748 g
= Mass of solvent (water) = 748 g


