
Pure copper may be produced by the reaction of copper(I) sulfide with oxygen gas as follows: Cu2S(s) + O2(g) 2Cu(s) + SO2(g) If 0.680 kg of copper(I) sulfide reacts with excess oxygen, what mass of copper metal may be produced ? A) 0.680 kg B) 0.136 kg C) 0.271 kg D) 0.543 kg E) 1.36 kg

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 16:30
Correct relationship between molecular formula and empirical formula
Answers: 1

Chemistry, 22.06.2019 17:00
The arrangement of particles is most ordered in a sample of
Answers: 1

Chemistry, 23.06.2019 01:20
How can parts of a solution be separated by chromatography?
Answers: 1

Chemistry, 23.06.2019 05:00
Which of the following describes qualitative data? a) recording the temperature of a solid as it is warmed. b) noting the color of a solution as it is heated. c) measuring the volume of an object by water displacement. d) taking the mass of an object using a balance.
Answers: 2
You know the right answer?
Pure copper may be produced by the reaction of copper(I) sulfide with oxygen gas as follows: Cu2S(s)...
Questions


Engineering, 01.09.2021 22:40


Physics, 01.09.2021 22:40




Mathematics, 01.09.2021 22:40




History, 01.09.2021 22:40

History, 01.09.2021 22:40

Physics, 01.09.2021 22:40

Mathematics, 01.09.2021 22:40


Mathematics, 01.09.2021 22:40

Mathematics, 01.09.2021 22:40


Biology, 01.09.2021 22:40

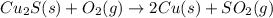
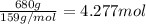
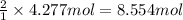 copper
copper

