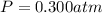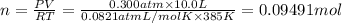Chemistry, 21.03.2020 02:59 carleygalloway103
0.100 mol of CaCO3 and 0.100 mol CaO are placed in an 10.0 L evacuated container and heated to 385 K. When equilibrium is reached the pressure of CO2 is 0.220 atm. 0.300 atm of CO2 is added, while keeping the temperature constant and the system is allowed to reach again equilibrium. What will be the final mass of CaCO3

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 10:30
Asample of air with a volume of 2.20m3 at a pressure of 105 kpa and a temperature of 30c is cooled to 10c and the pressure is reduced to 75.0 kpa. what is the new volume? 6.9 1.34 2.56 43.0 2.88
Answers: 1

Chemistry, 22.06.2019 14:20
You have a liquid that exhibits diltancy. you want to pour it from a bottle. what should you do to the bottle before pouring
Answers: 1


Chemistry, 22.06.2019 18:30
Asample of hydrated tin (ii) chloride (sncl2) has a mass of 4.90 g. when it is dehydrated, it has a mass of 4.10 g. which is the correct chemical formula for the hydrate? sncl2•2h2o sncl2•4h2o sncl2•6h2o
Answers: 2
You know the right answer?
0.100 mol of CaCO3 and 0.100 mol CaO are placed in an 10.0 L evacuated container and heated to 385 K...
Questions

History, 01.12.2021 08:20

History, 01.12.2021 08:20


History, 01.12.2021 08:20

Chemistry, 01.12.2021 08:20



English, 01.12.2021 08:20






Biology, 01.12.2021 08:20


Mathematics, 01.12.2021 08:20

Mathematics, 01.12.2021 08:20

World Languages, 01.12.2021 08:20


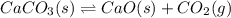
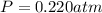
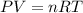 ( ideal gas equation)
( ideal gas equation)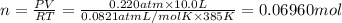
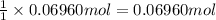 calcium carbonate
calcium carbonate