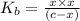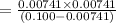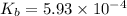Chemistry, 21.03.2020 03:01 bettybales1986
Aspirin (acetylsalicylic acid, C9H8O4) is a weak monoprotic acid. To determine its acid-dissociation constant, a student dissolved 2.00 g of aspirin in 0.600 L of water and measured the pH.
What was the Ka value calculated by the student if thepH of the solution was 2.62?
A 0.100 M solution of ethylamine (C2H5NH2) has a pH of 11.87.
Calculate the Kb for ethylamine.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 22:30
The diagram shows the relationship between scientific disciplines.the names of some scientific disciplines have been removed from the boxes. which scientific discipline belongs in the blue box? a.physics b.biology c.chemistry d.metallurgy
Answers: 2

Chemistry, 23.06.2019 09:00
What sources of error may have contributed to the percent yield not being 100 percent? think about things that may have led to inaccurate measurements or where mass of the product could have been lost if this experiment was conducted in a physical laboratory.
Answers: 2

Chemistry, 23.06.2019 09:20
1) a. water molecule breaks up into hydrogen and oxygen on passing electricity. does this involve breaking intermolecular or intramolecular forces of attraction. explain b. on boiling water changes to water vapor. does this involve breaking intermolecular or intramolecular forces of attraction. explain methanol evaporates faster than water. contrast the intermolecular forces and the vapor pressures of methanol and water?
Answers: 2

You know the right answer?
Aspirin (acetylsalicylic acid, C9H8O4) is a weak monoprotic acid. To determine its acid-dissociation...
Questions


Biology, 17.07.2019 12:30



Biology, 17.07.2019 12:30


Biology, 17.07.2019 12:30

Biology, 17.07.2019 12:30

Biology, 17.07.2019 12:30

Biology, 17.07.2019 12:30

Chemistry, 17.07.2019 12:30

Business, 17.07.2019 12:30

History, 17.07.2019 12:30

History, 17.07.2019 12:30

Computers and Technology, 17.07.2019 12:30





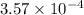 was the
was the 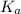 value calculated by the student.
value calculated by the student.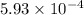 was the
was the 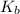 of ethylamine value calculated by the student.
of ethylamine value calculated by the student.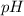 value of Aspirin solution = 2.62
value of Aspirin solution = 2.62![pH=-\log[H^+]](/tpl/images/0557/1305/cf945.png)
![[H^+]=10^{-2.62}=0.00240 M](/tpl/images/0557/1305/aef02.png)
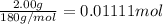
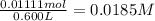
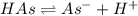
![K_a=\frac{[As^-][H^+]}{[HAs]}](/tpl/images/0557/1305/c8f9f.png) :
: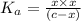
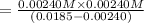
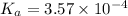
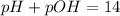
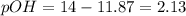
![pOH=-\log[OH^-]](/tpl/images/0557/1305/fe336.png)
![[OH^-]=10^{-2.13}=0.00741 M](/tpl/images/0557/1305/fa327.png)
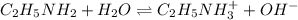
![K_b=\frac{[C_2H_5NH_3^{+}][OH^-]}{[C_2H_5NH_2]}](/tpl/images/0557/1305/63c9d.png) :
: