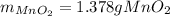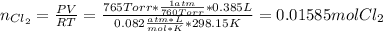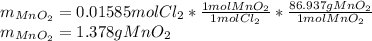Chlorine can be prepared in the laboratory by the reaction of manganese dioxide with hydrochloric acid, HCl(aq) , as described by the chemical equation
MnO2(s)+4HCl(aq)⟶MnCl2(aq)+2H2O(l)+ Cl2(g)
How much MnO2(s) should be added to excess HCl(aq) to obtain 385 mL Cl2(g) at 25 °C and 765 Torr ? mass of MnO 2

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 01:20
Match the acid base pairs by arranging the acid name with the conjugate base formula. hydrogen carbonate hydrogen phosphate carbonic acid read water sulfuric acid phosphoric acid a. co32- b. hso4- c. hco3- d. po43- e. h2po4- f. oh-
Answers: 1

Chemistry, 22.06.2019 03:30
Melting and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 2

Chemistry, 22.06.2019 14:10
Aconcentrated solution of ammonia is 14.8m and has a density of 0.899g/l. what is the concentration of ammonia in this solution in weight percent (%w/w)?
Answers: 1

You know the right answer?
Chlorine can be prepared in the laboratory by the reaction of manganese dioxide with hydrochloric ac...
Questions


Mathematics, 23.02.2021 01:00


Mathematics, 23.02.2021 01:00


English, 23.02.2021 01:00


Chemistry, 23.02.2021 01:00


Biology, 23.02.2021 01:00


Mathematics, 23.02.2021 01:00

Mathematics, 23.02.2021 01:00

Mathematics, 23.02.2021 01:00


History, 23.02.2021 01:00


Mathematics, 23.02.2021 01:00

Mathematics, 23.02.2021 01:00

Mathematics, 23.02.2021 01:00