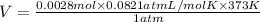It was important that the flask be completely dry before the unknown liquid was added so that water present would not vaporize when the flask was heated. A typical single drop of liquid water has a volume of approximately 0.050 mL. Assuming the density of liquid water is 1.00 g/mL, how many moles of water are in one drop of liquid, and what volume would this amount of water be?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 03:00
What happened in 2012 and how does it illustrate the importance of understanding the sun and how it works?
Answers: 3

Chemistry, 22.06.2019 15:50
Elements in group 2 are all called alkaline earth metals. what is most similar about the alkaline earth metals?
Answers: 1

Chemistry, 22.06.2019 19:30
What is the common name for the compound shown here? enter the common name of the compound shown?
Answers: 2

Chemistry, 22.06.2019 22:40
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization.a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution.part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1
You know the right answer?
It was important that the flask be completely dry before the unknown liquid was added so that water...
Questions


Physics, 20.08.2019 01:30

Geography, 20.08.2019 01:30


Mathematics, 20.08.2019 01:30


Mathematics, 20.08.2019 01:30


Engineering, 20.08.2019 01:30

Social Studies, 20.08.2019 01:30

Mathematics, 20.08.2019 01:30

History, 20.08.2019 01:30

Chemistry, 20.08.2019 01:30

Engineering, 20.08.2019 01:30

Mathematics, 20.08.2019 01:30





Physics, 20.08.2019 01:30

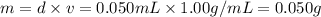
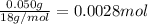
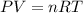 ( ideal gas equation )
( ideal gas equation )