
Chemistry, 24.03.2020 01:55 claudialopez093
The four major attractive forces between particles are ionic bonds, dipole-dipole attractions, hydrogen bonds, and dispersion forces. Consider the compounds below, and classify each by its predominant attractive or intermolecular force among atoms or molecules of the same type.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 08:00
Me i dont know what to do! the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1

Chemistry, 22.06.2019 14:30
For the reaction shown, find the limiting reactant for each of the following initial amounts of reactants. 4al(s)+3o2(g)→2al2o3(s) a) 1 molal, 1 mol o2 b) 4 molal, 2.6 mol o2 c) 16 molal, 13 mol o2 d) 7.4 molal, 6.5 mol o2
Answers: 3


Chemistry, 22.06.2019 23:00
What prefix multiplier is appropriate for reporting a measurement of 5.57 ×10−5 m?
Answers: 1
You know the right answer?
The four major attractive forces between particles are ionic bonds, dipole-dipole attractions, hydro...
Questions

English, 13.04.2021 20:10


Social Studies, 13.04.2021 20:10


Mathematics, 13.04.2021 20:10

Biology, 13.04.2021 20:10

History, 13.04.2021 20:10


Biology, 13.04.2021 20:10





Mathematics, 13.04.2021 20:10

Mathematics, 13.04.2021 20:10



Biology, 13.04.2021 20:10

Physics, 13.04.2021 20:10

Mathematics, 13.04.2021 20:10

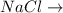 ionic bonds.
ionic bonds.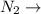 dispersion forces.
dispersion forces.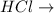 dipole-dipole attractions.
dipole-dipole attractions.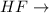 hydrogen bonds
hydrogen bonds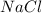 is formed between ions
is formed between ions 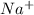 and
and 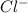 which makes it an ionic compound having ionic bonding between it's atoms.
which makes it an ionic compound having ionic bonding between it's atoms.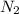 is a covalent non-polar molecule with only week dispersion forces caused by temporary dipoles in the binary molecule.
is a covalent non-polar molecule with only week dispersion forces caused by temporary dipoles in the binary molecule.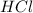 is a covalent polar molecule with permanent dipoles Hδ+ and Clδ-.
is a covalent polar molecule with permanent dipoles Hδ+ and Clδ-.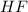 consists of both Hydrogen and Fluoride
consists of both Hydrogen and Fluoride 

