
Consider the reaction 2Na(s) + 2H2O(l) → 2NaOH(aq) + H2(g) When 2 moles of Na react with water at 25°C and 1 atm, the volume of H2 formed is 24.5 L. Calculate the magnitude of work done in joules when 0.55 g of Na reacts with water under the same conditions. (The conversion factor is 1 L · atm = 101.3 J.)

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 09:50
What are four significant sources of ghgs that come from wostem washington?
Answers: 2

Chemistry, 22.06.2019 11:00
Problem page combustion of hydrocarbons such as pentane ( c5 h12 ) produces carbon dioxide, a "greenhouse gas." greenhouse gases in the earth's atmosphere can trap the sun's heat, raising the average temperature of the earth. for this reason there has been a great deal of international discussion about whether to regulate the production of carbon dioxide.(a) write a balanced chemical equation, including physical state symbols, for the combustion of liquid pentane into gaseous carbon dioxide and gaseous water. (b) suppose 0.350 kg of pentane are burned in air at a pressure of exactly 1 atm and a temperature of 20.0 degree c. calculate the volume of carbon dioxide gas that is produced.be sure your answer has the correct number of significant digits.
Answers: 2

Chemistry, 22.06.2019 16:00
If 15 drops of ethanol from a medical dropper weight 0.60g, how many drops does it takes from a dropper to dispense 1.0ml of ethanol? the density of ethanol is 0.80g/ml
Answers: 1
You know the right answer?
Consider the reaction 2Na(s) + 2H2O(l) → 2NaOH(aq) + H2(g) When 2 moles of Na react with water at 25...
Questions


Mathematics, 15.05.2021 14:00


English, 15.05.2021 14:00

Mathematics, 15.05.2021 14:00




Mathematics, 15.05.2021 14:00

Computers and Technology, 15.05.2021 14:00


English, 15.05.2021 14:00

Advanced Placement (AP), 15.05.2021 14:00

Mathematics, 15.05.2021 14:00

Biology, 15.05.2021 14:00

English, 15.05.2021 14:00

Mathematics, 15.05.2021 14:00

Chemistry, 15.05.2021 14:00

Mathematics, 15.05.2021 14:00

Mathematics, 15.05.2021 14:00

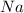
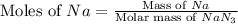
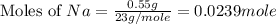
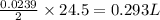 volume of hydrogen gas
volume of hydrogen gas

