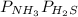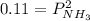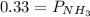Chemistry, 24.03.2020 16:55 ynclankaedon
Ammonium hydrogen sulfide NH4HS(s) decomposes on heating according to the reaction NH4HS(s) ↔ NH3(g) + H2S(g) At 25 °C the equilibrium cnstant of the reaction is Kp = 0.11. What is the total pressure at that temperature in a flask that was initially partially filled with a sizeable amount of NH4HS(s)?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:30
If 1.8 l of water is added to 2.5l of a 7.0 m koh solution, what is the molarity of the new solution
Answers: 1

Chemistry, 22.06.2019 15:10
The ozone molecule o3 has a permanent dipole moment of 1.8×10−30 cm. although the molecule is very slightly bent-which is why it has a dipole moment-it can be modeled as a uniform rod of length 2.5×10−10 m with the dipole moment perpendicular to the axis of the rod. suppose an ozone molecule is in a 8000 n/c uniform electric field. in equilibrium, the dipole moment is aligned with the electric field. but if the molecule is rotated by a small angle and released, it will oscillate back and forth in simple harmonic motion.what is the frequency f of oscillation?
Answers: 2

Chemistry, 22.06.2019 15:20
Select the most likely product for this reaction: koh(aq) + co2(g) – ? k2co3(aq) + h2o(1) k(s) + h2(g) + o2(g) k(s) + co3(9) +h2
Answers: 2

You know the right answer?
Ammonium hydrogen sulfide NH4HS(s) decomposes on heating according to the reaction NH4HS(s) ↔ NH3(g)...
Questions

Mathematics, 09.02.2021 05:50




Computers and Technology, 09.02.2021 05:50





History, 09.02.2021 05:50

Mathematics, 09.02.2021 05:50


Mathematics, 09.02.2021 05:50


Arts, 09.02.2021 05:50


History, 09.02.2021 05:50


English, 09.02.2021 05:50

Mathematics, 09.02.2021 05:50