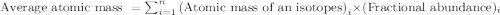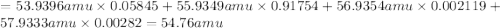Chemistry, 24.03.2020 17:02 ameliaxbowen65
Iron has many isotopes but only 4 are found in significant amounts in naturally found mixtures. The amounts by mass percent are: 5.845% of 54 Fe (53.9396 amu) 91.754% of 56Fe (55.9349 amu), 2.119% of 57Fe (56.9354 amu) and 0.282% of 58Fe (57.9333 amu). What would you determine the average mass of iron to be

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 10:00
Water's surface tension and heat storage capacity are accounted for by its a) orbitals b) weight c) hydrogen bonds d) mass e) size
Answers: 2

Chemistry, 22.06.2019 14:00
The two naturally occurring isotopes of chlorine are 35cl (34.969 amu, 75.77%) and 37cl (36.966 amu, 24.23%). the two naturally occurring isotopes of bromine are 79br (78.918 rm amu, 50.69%) and 81br (80.916 amu, 49.31%). chlorine and bromine combine to form bromine monochloride, brcl. 1. how many peaks will be present in a mass spectrum for brcl? the four combinations of molecule possible given these four isotopes are: 81br37cl, 81br35cl, 79br37cl, and 79br35cl. 2. what are the masses of the four different brcl molecules? express the masses using six significant figures, in decreasing numeric order (highest to lowest), separated by commas.
Answers: 3

Chemistry, 22.06.2019 21:00
Rays from the sun are not considered matter true or false
Answers: 2

You know the right answer?
Iron has many isotopes but only 4 are found in significant amounts in naturally found mixtures. The...
Questions

Mathematics, 11.10.2021 19:50


History, 11.10.2021 19:50

Mathematics, 11.10.2021 19:50



Geography, 11.10.2021 19:50

World Languages, 11.10.2021 19:50


Biology, 11.10.2021 19:50

Physics, 11.10.2021 19:50


Computers and Technology, 11.10.2021 19:50


Mathematics, 11.10.2021 19:50

Mathematics, 11.10.2021 19:50