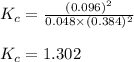Chemistry, 24.03.2020 17:01 emopandabogard8712
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. An industrial chemist studying this reaction fills a tank with of sulfur dioxide gas and of oxygen gas, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be . Calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. Round your answer to significant digits.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 04:30
Use the periodic table to determine the electron configuration of dysprosium (dy) and americium (am) in noble-gas notation.
Answers: 1

Chemistry, 22.06.2019 09:30
In 2002, the rare earth elements mine in mountain pass, california was closed because
Answers: 1


Chemistry, 22.06.2019 15:30
Which of the following are correct values for the ideal gas laws constant r
Answers: 1
You know the right answer?
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid...
Questions

History, 16.08.2019 20:10

English, 16.08.2019 20:10


English, 16.08.2019 20:10



Mathematics, 16.08.2019 20:10


English, 16.08.2019 20:10





Mathematics, 16.08.2019 20:10

English, 16.08.2019 20:10


English, 16.08.2019 20:10

English, 16.08.2019 20:10


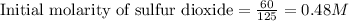
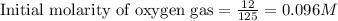
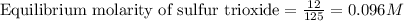
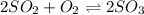
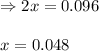
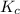 for above equation follows:
for above equation follows:![K_c=\frac{[SO_3]^2}{[O_2]\times [SO_2]^2}](/tpl/images/0560/9411/91096.png)