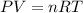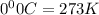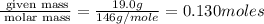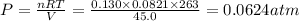Chemistry, 24.03.2020 17:37 connersitte1221l
A reaction between liquid reactants takes place at −10.0°C in a sealed, evacuated vessel with a measured volume of 45.0L. Measurements show that the reaction produced 19.g of sulfur hexafluoride gas. Calculate the pressure of sulfur hexafluoride gas in the reaction vessel after the reaction. You may ignore the volume of the liquid reactants. Be sure your answer has the correct number of significant digits.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 05:30
Transportation is the largest single source of air pollution in the united states. air pollution can harm the environment and human health. which technology could offer a solution to this problem? mufflers that reduce noise motors that run on electricity tires that improve gas mileage
Answers: 3


Chemistry, 23.06.2019 01:10
Volume is a measurement of how fast particles of a substance are moving
Answers: 3

Chemistry, 23.06.2019 01:20
Use the de broglie's wave equation to find the wavelength of an electron moving at 7.3 × 106 m/s. show your work. note: h = plank's constant (6.62607 x 10-34 j s)
Answers: 1
You know the right answer?
A reaction between liquid reactants takes place at −10.0°C in a sealed, evacuated vessel with a meas...
Questions

Social Studies, 16.10.2019 19:00

Physics, 16.10.2019 19:00



Mathematics, 16.10.2019 19:00


History, 16.10.2019 19:00

Social Studies, 16.10.2019 19:00

Mathematics, 16.10.2019 19:00


Biology, 16.10.2019 19:00



English, 16.10.2019 19:00


Chemistry, 16.10.2019 19:00

Mathematics, 16.10.2019 19:00

Biology, 16.10.2019 19:00


English, 16.10.2019 19:00