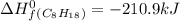For a particular isomer of C8H18, the combustion reaction produces 5113.3 kJ of heat per mole of C8H18(g) consumed, under standard conditions. C8H18(g)+252O2(g)⟶8CO2(g)+9H2O(g)ΔH ∘rxn=−5113.3 kJ/mol What is the standard enthalpy of formation of this isomer of C8H18(g)? ΔH∘f=

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 17:00
An aqueous solution of hydroiodic acid is standardized by titration with a 0.186 m solution of calcium hydroxide. if 26.5 ml of base are required to neutralize 20.3 ml of the acid, what is the molarity of the hydroiodic acid solution? m hydroiodic acid
Answers: 1

Chemistry, 22.06.2019 07:00
The boiling point of propanoic acid is higher than that of 1-butanol because: propanoic acid has a higher molecular weight than 1-butanol. propanoic acid is more soluble in water than 1-butanol. propanoic acid is a better hydrogen bond donor than 1-butanol. propanoic acid forms hydrogen bonded dimers and 1-butanol does not. 1-butanol forms hydrogen bonded dimers and propanoic acid does not.
Answers: 2

Chemistry, 22.06.2019 12:50
The number at the end of an isotope’s name is the number.
Answers: 1

Chemistry, 23.06.2019 02:30
What role does weathering have in shaping earth’s surface? a) it allows sediments to fall out of a medium. b) it sediments settle on a new surface. c) it breaks down older material into sediments. d) it transports sediments to a different location. will give brainliest, answer quickly.
Answers: 2
You know the right answer?
For a particular isomer of C8H18, the combustion reaction produces 5113.3 kJ of heat per mole of C8H...
Questions

Mathematics, 14.10.2019 03:00

Geography, 14.10.2019 03:00

Biology, 14.10.2019 03:00

Mathematics, 14.10.2019 03:00

Mathematics, 14.10.2019 03:00

Mathematics, 14.10.2019 03:00

Health, 14.10.2019 03:00

Computers and Technology, 14.10.2019 03:00

Social Studies, 14.10.2019 03:00

Mathematics, 14.10.2019 03:00

Physics, 14.10.2019 03:00


Mathematics, 14.10.2019 03:00





Chemistry, 14.10.2019 03:00

English, 14.10.2019 03:00

History, 14.10.2019 03:00

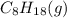 is -210.9 kJ
is -210.9 kJ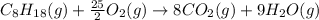
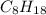 .
.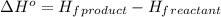
![\Delta H^o=[n_{CO_2}\times \Delta H_f^0_{(CO_2)}+n_{H_2O}\times \Delta H_f^0_{(H_2O)}]-[n_{O_2}\times \Delta H_f^0_{(O_2)+n_{C_8H_{18}}\times \Delta H_f^0_{(C_8H_{18})}]](/tpl/images/0561/1035/28ca9.png)
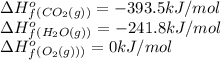
![-511.3kJ/mol=[(8\times -393.5)+(9\times -241.8)]-[(\frac{25}{2}\times 0)+(1\times \Delta H_f^0_{(C_8H_{18})}](/tpl/images/0561/1035/6b6c5.png)