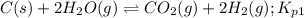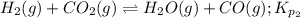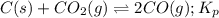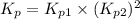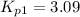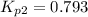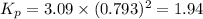Chemistry, 24.03.2020 21:33 Flamingmaster99
Calculate the value of K p for the equation C ( s ) + CO 2 ( g ) − ⇀ ↽ − 2 CO ( g ) K p = ? given that at a certain temperature C ( s ) + 2 H 2 O ( g ) − ⇀ ↽ − CO 2 ( g ) + 2 H 2 ( g ) K p 1 = 3.09 a t m H 2 ( g ) + CO 2 ( g ) − ⇀ ↽ − H 2 O ( g ) + CO ( g ) K p 2 = 0.793

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 12:20
The yearly amounts of carbon emissions from cars in belgium are normally distributed with a mean of 13.9 gigagrams per year and a standard deviation of 5.8 gigagrams per year. find the probability that the amount of carbon emissions from cars in belgium for a randomly selected year are between 11.5 gigagrams and 14.0 gigagrams per year. a. 0.340 b. 0.660 c. 0.167 d. 0.397
Answers: 2



Chemistry, 22.06.2019 16:00
What rule is used to determine how many covalent bonds an element can form? a. the number of covalent bonds is equal to six c the number of covalent bonds is equal to five minus the group number plus the group number b. the number of covalent bonds is equal to eight d. none of the above minus the group number select the best answer from the choices provided
Answers: 2
You know the right answer?
Calculate the value of K p for the equation C ( s ) + CO 2 ( g ) − ⇀ ↽ − 2 CO ( g ) K p = ? given th...
Questions

Mathematics, 09.07.2019 07:00

English, 09.07.2019 07:00

Health, 09.07.2019 07:00


Biology, 09.07.2019 07:00

Mathematics, 09.07.2019 07:00







Social Studies, 09.07.2019 07:00

Advanced Placement (AP), 09.07.2019 07:00



Social Studies, 09.07.2019 07:00

Biology, 09.07.2019 07:00

Biology, 09.07.2019 07:00

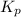 for the net reaction is 1.94
for the net reaction is 1.94