
The dimerization of butadiene 2C4H61 g2h C8H121 g2 was studied at 500. K, and the following data were obtained: Time (s) [C4H6] (mol/L) 195 1.6 3 1022 604 1.5 3 1022 1246 1.3 3 1022 2180 1.1 3 1022 6210 0.68 3 1022 Assuming that Rate 52 D3C4H64 Dt determine the form of the rate law, the integrated rate law, and the value of the rate constant for this reaction.

Answers: 3
Another question on Chemistry

Chemistry, 23.06.2019 00:50
What is the enthalpy of combustion (per mole) of c4h10 (g)? –2,657.5 kj/mol –5315.0 kj/mol –509.7 kj/mol –254.8 kj/mol
Answers: 1

Chemistry, 23.06.2019 01:00
What two factors can affect the properties of a hydrocarbon? a. the number of its carbon atoms and the number of single bonds b. the number of its carbon atoms and the arrangement of its atoms c. the arrangement of its atoms and the number of its double bonds
Answers: 1

Chemistry, 23.06.2019 02:50
Select the correct location on the image identify the element that humans need to breathe. 2015 er r ights reserved
Answers: 3

Chemistry, 23.06.2019 03:30
If you need to add 27.50ml of a solution, which piece of glassware would you use to deliver this volume and explain how you would determine if the 27.50 ml was measured?
Answers: 1
You know the right answer?
The dimerization of butadiene 2C4H61 g2h C8H121 g2 was studied at 500. K, and the following data wer...
Questions





Mathematics, 30.04.2021 17:20


Chemistry, 30.04.2021 17:20

World Languages, 30.04.2021 17:20

History, 30.04.2021 17:20

Mathematics, 30.04.2021 17:20

English, 30.04.2021 17:20



Mathematics, 30.04.2021 17:20

Mathematics, 30.04.2021 17:20


History, 30.04.2021 17:20

Business, 30.04.2021 17:20

Mathematics, 30.04.2021 17:20

History, 30.04.2021 17:20

![k[C_4H_6]^2](/tpl/images/0561/6934/d2fdb.png)
![\frac{1}{[C_4H_6]}=\frac{1}{[C_4H_6]_0}+kt](/tpl/images/0561/6934/38768.png)
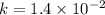
![\frac{1}{[C_4H_6]}](/tpl/images/0561/6934/fd07e.png) and the reaction is second order hence we get the rate law from
and the reaction is second order hence we get the rate law from ![k[A]^n](/tpl/images/0561/6934/3c428.png) .
. ![\frac{1}{[A]}=\frac{1}{[A]_0} +kt](/tpl/images/0561/6934/aad88.png) where A is
where A is 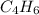 .
. 

