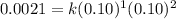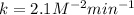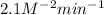The following data was collected for the formation of ammonia (NH3) based on the following overall reaction: N2 + 3H2 = 2NH3 N2 (M) H2 (M) Initial Rate (M/min.) 0.10 0.10 0.0021 0.10 0.20 0.0084 0.20 0.40 0.0672 What is the unit for the rate constant in the rate law for the formation of ammonia? Choose ONLY ONE and indicate your answer for this question on the exam answer sheet. a. M/min. b. 1/min

Answers: 2
Another question on Chemistry

Chemistry, 23.06.2019 00:00
The graph indicates the running route for tobias. which best describes his run? from time 0 to 6, he went fast and then slowed down. from time 6 to 10, he was at his slowest. from time 12 to 14, he went very slow. from time 14 to 18, he went toward the starting point.
Answers: 2

Chemistry, 23.06.2019 01:30
The solubility of barium nitrate is 9.02 g/100 g h2o at 20°c. a 15.2 g sample of barium nitrate is added to 200.0 g of water at 20°c. is the solution saturated, unsaturated, or supersaturated? a. unsaturated b. saturated c. supersaturated
Answers: 1

Chemistry, 23.06.2019 03:00
What happens in the particles of a gas when the gas is compressed
Answers: 1

Chemistry, 23.06.2019 15:40
Twenty-seven milliliters of an acid with an unknown concentration are titrated with a base that has a concentration of 0.55 m. the indicator changed color when 12.5 milliliters of base were added. what is the concentration of the unknown acid?
Answers: 2
You know the right answer?
The following data was collected for the formation of ammonia (NH3) based on the following overall r...
Questions

Physics, 06.06.2020 19:59

French, 06.06.2020 19:59




Mathematics, 06.06.2020 19:59



Chemistry, 06.06.2020 19:59




Social Studies, 06.06.2020 19:59

English, 06.06.2020 19:59


Advanced Placement (AP), 06.06.2020 19:59



Mathematics, 06.06.2020 19:59

English, 06.06.2020 19:59

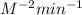
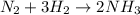
![\text{Rate}=k[N_2]^a[H_2]^b](/tpl/images/0562/1451/b6fd0.png)
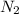
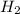
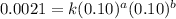 ....(1)
....(1)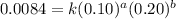 ....(2)
....(2)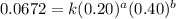 ....(3)
....(3)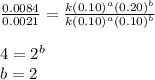
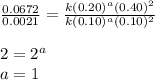
![\text{Rate}=k[N_2]^1[H_2]^2](/tpl/images/0562/1451/c46cd.png)