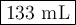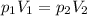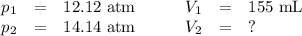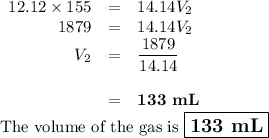Chemistry, 25.03.2020 05:02 ggdvj9gggsc
If i initially have a gas pressure of 12.12 atms, a volume of 155 ml, and a constant temperature of 200 degrees C, and then i raise the pressure to 14.14 atms, what is the volume of the gas?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 16:00
Use the table to identify the phase and phase changes of the elements under the given conditions. write the name of the substance, phase, or phase change. when the temperature in a room increases from 25°c to 33°c, changes from a solid to a liquid. in a lab, methane and nitrogen are cooled from -170°c to -200°c. the methane freezes and the nitrogen . when gold is heated to 2,856°c it changes from a liquid to a .
Answers: 2

Chemistry, 21.06.2019 17:00
Initially, the balloon had 3.0 liters of gas at a pressure of 400 kpa and was at a temperature of 294 k. if the balloon is cooled to 277 k and its volume decreased to 1 l, what will the new pressure in the balloon be?
Answers: 1

Chemistry, 22.06.2019 09:40
In the lab, ammonia was mixed with water to form ammonium hydroxide. what is/are the reactant(s)? o water and ammonia o ammonia o ammonium hydroxide need
Answers: 2

Chemistry, 22.06.2019 10:00
3. how much energy in joules is required to evaporate .0005 kg of liquid ammonia to vapor at the same temperature? 4. how much energy ( in megajoules ) is given up by .75 kg of water at 0c when it freezes to form ice at 0c? 5. explain how heat works between and at critical temperatures?
Answers: 2
You know the right answer?
If i initially have a gas pressure of 12.12 atms, a volume of 155 ml, and a constant temperature of...
Questions


History, 15.01.2020 05:31


Engineering, 15.01.2020 05:31




Mathematics, 15.01.2020 05:31

Mathematics, 15.01.2020 05:31



Mathematics, 15.01.2020 05:31

Mathematics, 15.01.2020 05:31

Computers and Technology, 15.01.2020 05:31

Computers and Technology, 15.01.2020 05:31