Chemistry, 25.03.2020 05:41 alexisthegirl
What volume of carbon dioxide will 3.26 g of antacid made of calcium carbonate produce at 37.0 °C and 1.00 atm in the stomach according to the following reaction?
CaCO₃ (s) + 2 HCl (aq) → CaCl₂ (aq) + H₂O (l) + CO₂ (g)

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 03:30
If you have 5.25 grams of methane (ch4), how many grams of co2 will you produce ?
Answers: 1

Chemistry, 22.06.2019 10:00
Part 1: include important facts found through your research. part 2: include your visual display. include your summary of “the chemistry of water” from the national science foundation website. include your experiment. part 3: include responses to the reflection questions.
Answers: 1

Chemistry, 22.06.2019 11:30
Aperfume bottle is dropped in the corner of a room. the odor of the perfume can be detected on the other side of the room. which statement best describes this observation?
Answers: 2

Chemistry, 22.06.2019 23:00
What is the solubility-product constant of barium sulfate, baso4, if a saturated solution is 1.03 ´ 10-5 m?
Answers: 3
You know the right answer?
What volume of carbon dioxide will 3.26 g of antacid made of calcium carbonate produce at 37.0 °C an...
Questions




Mathematics, 07.01.2021 02:30

Spanish, 07.01.2021 02:30

Mathematics, 07.01.2021 02:30

Spanish, 07.01.2021 02:30

Chemistry, 07.01.2021 02:30





Mathematics, 07.01.2021 02:30


Mathematics, 07.01.2021 02:30

Social Studies, 07.01.2021 02:30




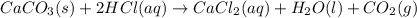
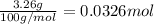
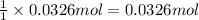 of carbon dioxide.
of carbon dioxide.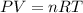 ( ideal gas equation )
( ideal gas equation )