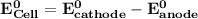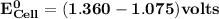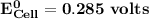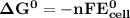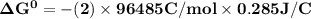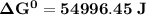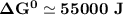Chemistry, 25.03.2020 05:26 kenzie3497
Determine ΔG° for a cell that utilizes the following reaction: Cl2(g) + 2Br–(aq) → 2Cl–(aq) + Br2(l) The standard reduction for the chlorine gas is 1.360 volts and the standard reduction for the bromine liquid is about 1.075 volts. Group of answer choices

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 02:10
When 225mg of anthracene, c14h10(s), was burned in a bomb calorimeter the temperature rose by 1.75k. calculate the calorimeter constant. by how much will the temperature rise when 125mg of phenol, c6h5oh(s), is burned in the calorimeter under the same conditions? (δch< (c14h10,s)=–7061 kj mol−1.)
Answers: 3

Chemistry, 22.06.2019 21:30
Which of the following changes will decrease the total amount of gaseous solute able to be dissolved in a liter of liquid water? (2 points) decreasing temperature decreasing pressure decreasing surface area decreasing solute concentration
Answers: 1

Chemistry, 22.06.2019 22:30
What is the value of the standard enthalpy of formation of an element in its most stable form?
Answers: 3

Chemistry, 23.06.2019 01:30
What is produced from neutralization of an acid and a base? a. hydronium ions b. citric acid c. salt and water
Answers: 1
You know the right answer?
Determine ΔG° for a cell that utilizes the following reaction: Cl2(g) + 2Br–(aq) → 2Cl–(aq) + Br2(l)...
Questions



Mathematics, 06.03.2021 02:10

Mathematics, 06.03.2021 02:10


Computers and Technology, 06.03.2021 02:10

Mathematics, 06.03.2021 02:10

Mathematics, 06.03.2021 02:10

History, 06.03.2021 02:10



Mathematics, 06.03.2021 02:10






Mathematics, 06.03.2021 02:10

Mathematics, 06.03.2021 02:10

Social Studies, 06.03.2021 02:10

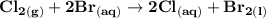
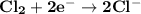
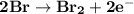
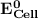 can be calculated as:
can be calculated as: