Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 23:30
Calculate the expected ph values of the buffer systems from the experiments (a,b,c,d), using the henderson- hasselbalch equation, ph-pka+log[a-]/[ha]. use for pka values carbonic acid= 6.37, and acetic acid= 4.75.
Answers: 2

Chemistry, 22.06.2019 09:30
In apex! a liquid heated beyond a certain temperature becomes
Answers: 1

Chemistry, 22.06.2019 12:00
In a laboratory, 1.55mg of an organic compound containing carbon, hydrogen, and oxygen is burned for analysis. this combustion resulted in the formation of 1.45mg of carbon dioxide and .89 mg of water. what is the empirical formula for this compound?
Answers: 1

Chemistry, 22.06.2019 18:10
The atom fluorine generally will become stable through the formation of an ionic chemical compound by accepting electron(s) from another atom. this process will fill its outer energy level of electrons.
Answers: 1
You know the right answer?
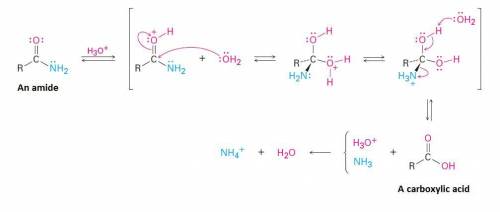
With the correct choice of acid, acid hydrolysis of ethanamide could produce A) ethanoic acid and am...
Questions


Mathematics, 27.06.2019 07:50

Mathematics, 27.06.2019 07:50

Biology, 27.06.2019 07:50

History, 27.06.2019 07:50




History, 27.06.2019 07:50

Biology, 27.06.2019 07:50


Physics, 27.06.2019 07:50


Mathematics, 27.06.2019 07:50


Mathematics, 27.06.2019 07:50


History, 27.06.2019 07:50

Physics, 27.06.2019 07:50