
Chemistry, 25.03.2020 16:17 jimarieb08
In Part A, you found the number of moles of product (1.80 mol P2O5 ) formed from the given amount of phosphorus and excess oxygen. In Part B, you found the number of moles of product (1.40 mol P2O5 ) formed from the given amount of oxygen and excess phosphorus. Now, determine the number of moles of P2O5 is produced from the given amounts of phosphorus and oxygen

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:00
Your friend offers to show you an intrusive igneous rock. which of the following would you expect to see?
Answers: 1

Chemistry, 21.06.2019 22:30
Agas at 155 kpa and standard temperature has an initial volume of 1.00 l. the pressure of the gas rises to 500 kpa as the temperature also rises to 135°c. what is the new volume? 2.16 l 0.463 l 0.207 l 4.82 l
Answers: 3

Chemistry, 22.06.2019 11:40
Which of these expressions are correct variations of the combined gas law? p1v1t2 = p2v2t1 both
Answers: 2

Chemistry, 22.06.2019 15:00
Why does a plastic bottle that is sealed at a high altitude change it’s shape when taken to lower altitude
Answers: 2
You know the right answer?
In Part A, you found the number of moles of product (1.80 mol P2O5 ) formed from the given amount of...
Questions

Mathematics, 21.12.2020 14:40

Business, 21.12.2020 14:40



Mathematics, 21.12.2020 14:40


Mathematics, 21.12.2020 14:40

German, 21.12.2020 14:40


Biology, 21.12.2020 14:40

Mathematics, 21.12.2020 14:50

History, 21.12.2020 14:50


Mathematics, 21.12.2020 14:50

English, 21.12.2020 14:50



Mathematics, 21.12.2020 14:50

Mathematics, 21.12.2020 14:50

Law, 21.12.2020 14:50

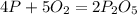
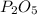 is formed by 4 moles of
is formed by 4 moles of 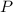
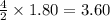
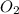 required =
required = 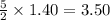
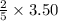 = 1.40 moles.
= 1.40 moles.

