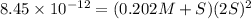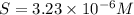Chemistry, 26.03.2020 02:53 erbnichole
Consider the dissolution of AB(s): AB(s)⇌A+(aq)+B−(aq) Le Châtelier's principle tells us that an increase in either [A+] or [B−] will shift this equilibrium to the left, reducing the solubility of AB. In other words, AB is more soluble in pure water than in a solution that already contains A+ or B− ions. This is an example of the common-ion effect. The generic metal hydroxide M(OH)2 has Ksp = 8.45×10−12. (NOTE: In this particular problem, because of the magnitude of the Ksp and the stoichiometry of the compound, the contribution of OH− from water can be ignored. However, this may not always be the case.) Part A What is the solubility of M(OH)2 in pure water? Express your answer with the appropriate units. View Available Hint(s) nothing nothing Part B What is the solubility of M(OH)2 in a 0.202 M solution of M(NO3)2? Express your answer with the appropriate units. View Available Hint(s) nothing nothing

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 13:00
These questions are based on the attached photo. the experiment is about burning magnesium metal with oxygen. 1. write the balanced chemical equation for the reaction you are performing. 2. calculate the mass of magnesium metal used in each trial. o trial 1: o trial 2: 3. calculate the actual yield of magnesium oxide for each trial. o trial 1: o trial 2: 4. magnesium is the limiting reactant in this experiment. calculate the theoretical yield of mgo for each trial. o trial 1: o trial 2: 5. determine the percent yield of mgo for your experiment for each trial. o trial 1: o trial 2: 6. determine the average percent yield of mgo for the two trials. your company currently uses a process with a similar cost of materials that has an average percent yield of 91 percent. if the average percent yield of this process is higher than that, this could save the company money. what is your recommendation to the company? support your recommendation using your data, calculations, and understanding of stoichiometry gathered from this lab.
Answers: 1

Chemistry, 23.06.2019 06:30
Acompound has the molecular formula c3h8. which class of organic compounds does it belong to?
Answers: 1

Chemistry, 23.06.2019 06:40
8. how much enthalpy/heat is transferred when 0.5113gof ammonia (nh3) reacts with excess oxygen according| to the following equation: 4nh3 +502 - 4n0+ 6h20ah = -905.4j
Answers: 1
You know the right answer?
Consider the dissolution of AB(s): AB(s)⇌A+(aq)+B−(aq) Le Châtelier's principle tells us that an inc...
Questions



Mathematics, 14.10.2019 19:30




History, 14.10.2019 19:30

Social Studies, 14.10.2019 19:30



Geography, 14.10.2019 19:30

Mathematics, 14.10.2019 19:30

History, 14.10.2019 19:30


History, 14.10.2019 19:30

Advanced Placement (AP), 14.10.2019 19:30

English, 14.10.2019 19:30

Social Studies, 14.10.2019 19:30


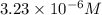 is the solubility of
is the solubility of 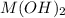 in a 0.202 M solution of
in a 0.202 M solution of 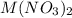 .
.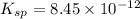
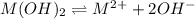
![K_{sp}=[M^{2+}][OH^-]^2](/tpl/images/0564/5689/a461b.png)
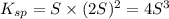
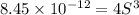
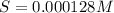
![[M^{2+}]=0.202 M+S](/tpl/images/0564/5689/48853.png)