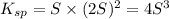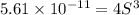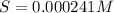Chemistry, 26.03.2020 23:46 tinasidell1972
Mg(OH)2 is a sparingly soluble compound, in this case a base, with a solubility product, Ksp, of 5.61×10−11. It is used to control the pH and provide nutrients in the biological (microbial) treatment of municipal wastewater streams. Based on the given value of the Ksp, what is the molar solubility of Mg(OH)2 in pure H2O?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 02:00
Which of the following happens during cell division? (a) energy is created (b) waste is eliminated (c) carbon dioxide is released (d) damaged cells are replaced
Answers: 1

Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2

Chemistry, 22.06.2019 09:00
Scientific evidence tells us that the cause of earths four season is the tilt of earth as it revolves around the sun. the student is instructed to illustrate this information in a science notebook. how will the student illiterate winter in the northern hemisphere?
Answers: 3

Chemistry, 22.06.2019 16:00
As changes in energy levels of electrons increase, the frequencies of atomic line spectra they emit
Answers: 2
You know the right answer?
Mg(OH)2 is a sparingly soluble compound, in this case a base, with a solubility product, Ksp, of 5.6...
Questions

Mathematics, 27.01.2020 20:31


Social Studies, 27.01.2020 20:31


Mathematics, 27.01.2020 20:31

Mathematics, 27.01.2020 20:31


History, 27.01.2020 20:31



Computers and Technology, 27.01.2020 20:31




Arts, 27.01.2020 20:31

Computers and Technology, 27.01.2020 20:31



Computers and Technology, 27.01.2020 20:31

Computers and Technology, 27.01.2020 20:31

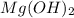 in pure water.
in pure water.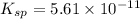
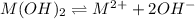
![K_{sp}=[M^{2+}][OH^-]^2](/tpl/images/0566/2963/a461b.png)