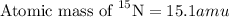The element nitrogen has an atomic weight of 14.0 and consists of two stable isotopes nitrogen-14 and nitrogen-15. The isotope nitrogen-14 has a mass of 14.0 amu and a percent natural abundance of 99.6 %. The isotope nitrogen-15 has a percent natural abundance of 0.370 %. What is the mass of nitrogen-15? amu

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 14:30
Clouds form when water vapor to form small droplets. a. humidifies b. condenses c. evaporates d. precipitates
Answers: 2


Chemistry, 22.06.2019 12:10
If a molecule with a molecular formula of c13h18 is treated with an excess of h2 in the presence of finally divided pt metal under conditions required for maximum hydrogenation of the molecule to give a molecule with a formula c13h24, how many rings are in the molecule?
Answers: 3

Chemistry, 22.06.2019 21:20
40dm3 of gas at 760 torr are heated from 5°c to 50°c what is the new volume
Answers: 3
You know the right answer?
The element nitrogen has an atomic weight of 14.0 and consists of two stable isotopes nitrogen-14 an...
Questions



Mathematics, 09.06.2021 19:10

English, 09.06.2021 19:10

Mathematics, 09.06.2021 19:10


Mathematics, 09.06.2021 19:10



Mathematics, 09.06.2021 19:10

Mathematics, 09.06.2021 19:10


Biology, 09.06.2021 19:10

History, 09.06.2021 19:10


Mathematics, 09.06.2021 19:10

Mathematics, 09.06.2021 19:10

Arts, 09.06.2021 19:10


Mathematics, 09.06.2021 19:10

 .....(1)
.....(1)
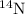 isotope:
isotope: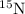 isotope:
isotope:![14.0=[(14.0\times 0.996)+(\text{Atomic mass of }^{15}\textrm{N}\times 0.00370)]](/tpl/images/0566/2813/d6364.png)