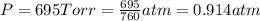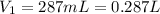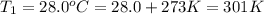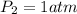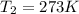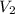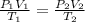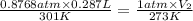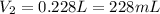Chemistry, 27.03.2020 01:04 slycooper99
A cylinder of compressed gas rolls off a boat and falls to the bottom of a lake. Eventually it rusts and the gas bubbles to the surface. A chemist collects a sample of the gas with the idea of trying to identify the gas. The wet gas collected occupies a volume of 287 mL at a pressure of 695 torr and temperature of 28.0oC. The vapor pressure of water at 28.0oC is 0.0372 atm. 1. Calculate the volume (L) that the gas occupies after it is dried (the water vapor removed) and stored at STP.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 18:00
The compound methyl butanoate smells like apples. its percent composition is 58.8% c, 9.9% h, and 31.4% o. what’s the empirical formula ?
Answers: 1


Chemistry, 22.06.2019 11:00
What is the molar mass of a gas that has density of 2.054 g/l
Answers: 2

Chemistry, 22.06.2019 11:00
Surface currents are caused by blank space . question 14 options: surface currents are caused by? differences in water temperature high salinity differences in density wind forces
Answers: 1
You know the right answer?
A cylinder of compressed gas rolls off a boat and falls to the bottom of a lake. Eventually it rusts...
Questions


Mathematics, 18.04.2020 00:01


Mathematics, 18.04.2020 00:01


Mathematics, 18.04.2020 00:01




Mathematics, 18.04.2020 00:01

Biology, 18.04.2020 00:01

Mathematics, 18.04.2020 00:01







Geography, 18.04.2020 00:01

Mathematics, 18.04.2020 00:01