Chemistry, 27.03.2020 18:09 aaronolivera200161
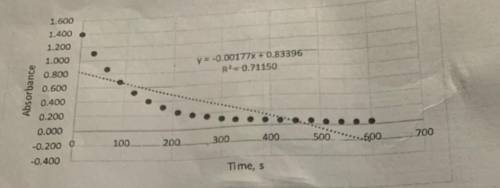
Determine the value of k' from the slope. These graphs were plotted in terms of absorbance, but the rate constant should be in terms of concentration. Beer-Lambert's law and the literature value for the molar absorptivity constant (87,000 M^-1cm^-1 can be used to convert the slope from units of absorbance/s to units of concentration/s. This value is the pseudo rate constant, k'.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 16:20
When water dissolves sugar, which process is not involved? o dissociation o hydration o surface area of the solute increases sa
Answers: 1

Chemistry, 22.06.2019 22:30
Which of the following is true about the speed of light? it depends on the wavelength.
Answers: 3

Chemistry, 23.06.2019 03:00
What does a complete balanced chemical equation include? a. exothermic coefficients b. endothermic coefficients c. valence electrons d. molar coefficients
Answers: 1
You know the right answer?
Determine the value of k' from the slope. These graphs were plotted in terms of absorbance, but the...
Questions







Biology, 07.03.2020 00:14



Biology, 07.03.2020 00:14




Mathematics, 07.03.2020 00:14





Mathematics, 07.03.2020 00:14

Computers and Technology, 07.03.2020 00:14