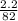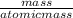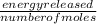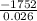A calorimeter contains 30.0 mL of water at 12.5 ∘C . When 2.20 g of X (a substance with a molar mass of 82.0 g/mol ) is added, it dissolves via the reaction X(s)+H2O(l)→X(aq) and the temperature of the solution increases to 25.5 ∘C . Calculate the enthalpy change, ΔH, for this reaction per mole of X. Assume that the specific heat of the resulting solution is equal to that of water [4.18 J/(g⋅∘C)], that density of water is 1.00 g/mL, and that no heat is lost to the calorimeter itself, nor to the surroundings

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 17:00
Astable electron arrangement for an atom is one that does not easily change. how is this arrangement arrived at? a. valence electrons are transferred or shared to create a full outer shell of electrons. b. valence electrons are discarded into space to create a full outer shell of electrons. c. protons (positive charge) pair with valence electrons (negative charge) to create a strong bond. d. outer shells with valence electrons are transferred or shared.
Answers: 2

Chemistry, 22.06.2019 19:00
Which change to the system wood cause the freely-moving piston to lower?
Answers: 1

Chemistry, 22.06.2019 21:50
If e is the symbol for an element, which two of the following symbols represent isotopes of the same element? 1. e2. e3. ea.1 and 2c.1 and 4b.3 and 4d.2 and 3
Answers: 2
You know the right answer?
A calorimeter contains 30.0 mL of water at 12.5 ∘C . When 2.20 g of X (a substance with a molar mass...
Questions

Biology, 27.08.2019 23:30

Geography, 27.08.2019 23:30

Mathematics, 27.08.2019 23:30



Physics, 27.08.2019 23:30


Mathematics, 27.08.2019 23:30


Mathematics, 27.08.2019 23:30

Social Studies, 27.08.2019 23:30

Mathematics, 27.08.2019 23:30





Mathematics, 27.08.2019 23:30

Social Studies, 27.08.2019 23:30