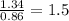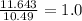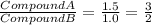Chemistry, 30.03.2020 16:16 kenishawilkinsoy4mgw
Sulfur reacts with oxygen and creates two compounds. Compound A contains 1.34 g of sulfur for every 0.86 g of oxygen. Compound B contains 11.63 g of sulfur for every 10.49 g of oxygen. What is the mass ratio of oxygen rounded to the nearest whole number

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 04:30
In which phase(s) do the molecules take the shape of the container?
Answers: 1

Chemistry, 22.06.2019 12:10
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3


Chemistry, 22.06.2019 12:30
Suppose you wanted to make 100 grams of water. what is the molar mass of water (h2o)?
Answers: 2
You know the right answer?
Sulfur reacts with oxygen and creates two compounds. Compound A contains 1.34 g of sulfur for every...
Questions

Mathematics, 20.10.2019 07:10



Mathematics, 20.10.2019 07:10

Chemistry, 20.10.2019 07:10

Mathematics, 20.10.2019 07:10



Mathematics, 20.10.2019 07:10





History, 20.10.2019 07:10

English, 20.10.2019 07:10


Mathematics, 20.10.2019 07:10

Mathematics, 20.10.2019 07:10

Mathematics, 20.10.2019 07:10