
Chemistry, 30.03.2020 17:01 zachtsmith2001
Be sure to answer all parts. Hydrogen iodide decomposes according to the reaction 2 HI(g) ⇌ H2(g) + I2(g) A sealed 1.50−L container initially holds 0.00623 mol of H2, 0.00414 mol of I2, and 0.0244 mol of HI at 703 K. When equilibrium is reached, the concentration of H2(g) is 0.00467 M. What are the concentrations of HI(g) and I2(g)?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 15:30
A1.5l container holds p.50 grams of an unknown gas at a pressure of 0.44 atm and a temperature of 50.c what is the molar mass of the unknown gas
Answers: 1

Chemistry, 22.06.2019 23:00
How does the value of the equilibrium constant show that a reaction reaches equilibrium very quickly? (a) the equilibrium constant is large. (b) the equilibrium constant is small. (c) the equilibrium constant is zero. (d) the value of the equilibrium constant does not show how quickly a reaction comes to equilibrium.
Answers: 1

Chemistry, 22.06.2019 23:30
The density of the solid phase of a substance is 0.90 g/cm3 and the density of the liquid phase is 1.0 g/cm3. a large increase in pressure will a. lower the freezing point b. raise the freezing point c. lower the boiling point d. raise the triple point e. lower the triple point
Answers: 1

Chemistry, 23.06.2019 06:30
Aplanet similar to earth has four moons roughly the same distance away. the moon that will most affect tides on the planet is the one that has the greatest a) mass. b) volume. c) density. d) amount of water.
Answers: 1
You know the right answer?
Be sure to answer all parts. Hydrogen iodide decomposes according to the reaction 2 HI(g) ⇌ H2(g) +...
Questions

Biology, 19.01.2020 08:31


Biology, 19.01.2020 08:31



History, 19.01.2020 08:31

Mathematics, 19.01.2020 08:31

Health, 19.01.2020 08:31



Social Studies, 19.01.2020 08:31






Biology, 19.01.2020 08:31

Mathematics, 19.01.2020 08:31

Spanish, 19.01.2020 08:31

History, 19.01.2020 08:31

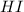 and
and 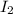 at equilibrium is, 0.0158 M and 0.00302 M respectively.
at equilibrium is, 0.0158 M and 0.00302 M respectively.
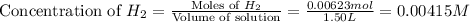
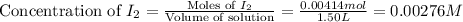
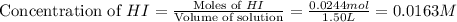
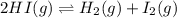
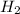 at equilibrium = 0.00467 M
at equilibrium = 0.00467 M

