
Chemistry, 30.03.2020 17:18 yesman1288
Consider: CO(g) + Cl2 (g) ⇌ COCl2 (g) Kc = 1.2×103 at 395 °C. If the equilibrium concentrations of Cl2 and COCl2 are the same at 395 °C, find the equilibrium concentration of CO in the reaction.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 21:40
It is important to wear proper protective equipment in lab even when not actively performing experiments because accidents can affect any researcher, even one not working on an experiment. select the best answer from the choices provided
Answers: 3

Chemistry, 22.06.2019 00:10
According to the diagram; a) identify the anode of the cell and write the half-reaction that occurs there b) write the overall equation for the reaction that occurs as the cell operates c) calculate the value of the standard cell potential ,e cell. d)write the shorthand notation of the cell above e)indicate the flow of the electrons on the diagram
Answers: 3

Chemistry, 22.06.2019 03:40
Kc = 0.040 for the system below at 450oc. if a reaction is initiated with 0.40 mole of cl2 and 0.40 mole of pcl3 in a 2.0 liter container, what is the equilibrium concentration of cl2 in the same system? pcl5(g) ⇄ pcl3(g) + cl2(g)
Answers: 3

You know the right answer?
Consider: CO(g) + Cl2 (g) ⇌ COCl2 (g) Kc = 1.2×103 at 395 °C. If the equilibrium concentrations of C...
Questions


Mathematics, 16.12.2020 16:50


Physics, 16.12.2020 16:50





Mathematics, 16.12.2020 16:50







Computers and Technology, 16.12.2020 16:50




Mathematics, 16.12.2020 16:50

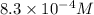
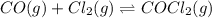
![K_c=\frac{[COCl_2]}{[CO][Cl_2]}](/tpl/images/0570/4835/36d91.png)
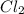 at equilibrium = Concentration of
at equilibrium = Concentration of 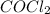
![K_c=\frac{[Cl_2]}{[CO][Cl_2]}](/tpl/images/0570/4835/18881.png)
![K_c=\frac{1}{[CO]}](/tpl/images/0570/4835/6d26f.png)
![1.2\times 10^3=\frac{1}{[CO]}](/tpl/images/0570/4835/d1c27.png)
![[CO]=8.3\times 10^{-4}M](/tpl/images/0570/4835/cb9b9.png)


