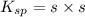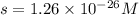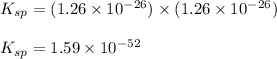Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 14:50
Given the following information: mass of proton = 1.00728 amu mass of neutron = 1.00866 amu mass of electron = 5.486 × 10^-4 amu speed of light = 2.9979 × 10^8 m/s calculate the nuclear binding energy (absolute value) of 3li^6. which has an atomic mass of 6.015126 amu. j/mol.
Answers: 2

Chemistry, 22.06.2019 20:00
What happens to the temperature of a substance when the average kinetic energy of its particles increases?
Answers: 3

Chemistry, 22.06.2019 20:30
We are hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 3
You know the right answer?
At 25 oC the solubility of mercury(II) sulfide is 1.26 x 10-26 mol/L. Calculate the value of Ksp at...
Questions







Mathematics, 23.02.2021 08:30


Social Studies, 23.02.2021 08:30



English, 23.02.2021 08:30

Mathematics, 23.02.2021 08:30

Mathematics, 23.02.2021 08:30

History, 23.02.2021 08:30

Mathematics, 23.02.2021 08:30




Biology, 23.02.2021 08:30

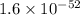
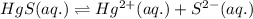
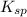 for above equation follows:
for above equation follows: