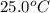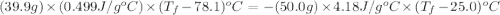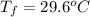A hot lump of 39.9 g of iron at an initial temperature of 78.1 °C is placed in 50.0 mL H 2 O initially at 25.0 °C and allowed to reach thermal equilibrium. What is the final temperature of the iron and water, given that the specific heat of iron is 0.449 J/(g⋅°C)? Assume no heat is lost to surroundings.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 12:10
If a molecule with a molecular formula of c13h18 is treated with an excess of h2 in the presence of finally divided pt metal under conditions required for maximum hydrogenation of the molecule to give a molecule with a formula c13h24, how many rings are in the molecule?
Answers: 3

Chemistry, 22.06.2019 14:00
What is the ph of a solution that has a hydrogen ion concentration of 1.0 * 10 -9 m?
Answers: 2

Chemistry, 22.06.2019 20:00
Which of the following would not diffuse through the plasma membrane by means of simple diffusion? 1 oxygen 2 glucose 3 a steroid hormone 4 a lipid soluble vitamin
Answers: 3

Chemistry, 23.06.2019 01:00
If a straight-chain hydrocarbon is a gas at room temperature, how many carbon atoms will it have? a. 6 carbon atoms b. 12 carbon atoms c. 24 carbon atoms d. 3 carbon atoms
Answers: 1
You know the right answer?
A hot lump of 39.9 g of iron at an initial temperature of 78.1 °C is placed in 50.0 mL H 2 O initial...
Questions

Mathematics, 31.12.2019 15:31


Advanced Placement (AP), 31.12.2019 15:31

Mathematics, 31.12.2019 15:31

Mathematics, 31.12.2019 15:31

Mathematics, 31.12.2019 15:31

Chemistry, 31.12.2019 15:31




Mathematics, 31.12.2019 15:31




Mathematics, 31.12.2019 15:31

Mathematics, 31.12.2019 15:31


Physics, 31.12.2019 15:31

Biology, 31.12.2019 15:31

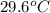
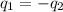
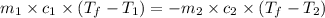
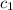 = specific heat of iron =
= specific heat of iron = 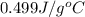
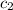 = specific heat of water =
= specific heat of water = 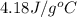
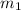 = mass of iron = 39.9 g
= mass of iron = 39.9 g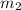 = mass of water =
= mass of water = 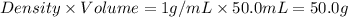
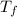 = final temperature of mixture = ?
= final temperature of mixture = ?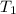 = initial temperature of iron =
= initial temperature of iron = 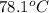
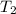 = initial temperature of water =
= initial temperature of water =