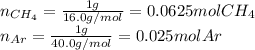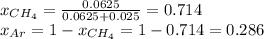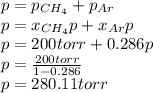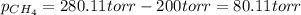Chemistry, 30.03.2020 19:32 bvbbridesmaid5519
A gas mixture consists of equal masses of methane (molecular weight 16.0) and argon (atomic weight 40.0). If the partial pressure of argon is 200. torr, what is the pressure of methane, in torr

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 23:00
Matches the chemical name of each oxide of phosphorus to its chemical formula
Answers: 2


Chemistry, 22.06.2019 15:30
A1.5l container holds p.50 grams of an unknown gas at a pressure of 0.44 atm and a temperature of 50.c what is the molar mass of the unknown gas
Answers: 1

You know the right answer?
A gas mixture consists of equal masses of methane (molecular weight 16.0) and argon (atomic weight 4...
Questions





Mathematics, 25.02.2020 01:37





Mathematics, 25.02.2020 01:37

Mathematics, 25.02.2020 01:37



Mathematics, 25.02.2020 01:37





History, 25.02.2020 01:38