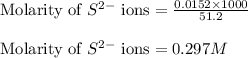Chemistry, 30.03.2020 19:28 kylieweeks052704
In the laboratory a student combines 23.5 mL of a 0.173 M barium sulfide solution with 27.7 mL of a 0.402 M sodium sulfide solution. What is the final concentration of sulfide anion ?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 20:30
10. translate each of the following chemical equations into a sentence. a. 2 zns(s) + 3 o2(g) -> 2 zno(s) + 2 so2(g) b. cah2(s) + 2 h2o(l) -> ca(oh)2 (aq) + 2 h2(g)
Answers: 2

Chemistry, 22.06.2019 05:30
Which of the following signs of a chemical reaction are observed in the reaction of potassium with water? precipitate formed temperature change smell produced gas produced color change
Answers: 2

Chemistry, 22.06.2019 06:30
Predict whether the changes in enthalpy, entropy, and free energy will be positive or negative for the boiling of water, and explain your predictions. how does temperature affect the spontaneity of this process?
Answers: 1

Chemistry, 22.06.2019 20:30
Calculate the percent composition by mass of each element in al(oh)3. use at least three significant figures.
Answers: 1
You know the right answer?
In the laboratory a student combines 23.5 mL of a 0.173 M barium sulfide solution with 27.7 mL of a...
Questions

English, 19.03.2021 03:00


Mathematics, 19.03.2021 03:00

Mathematics, 19.03.2021 03:00

Mathematics, 19.03.2021 03:00


Chemistry, 19.03.2021 03:00

Health, 19.03.2021 03:00






History, 19.03.2021 03:00

Mathematics, 19.03.2021 03:00




English, 19.03.2021 03:00

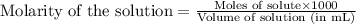 .....(1)
.....(1)
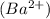 and 1 mole of sulfide ions
and 1 mole of sulfide ions 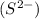
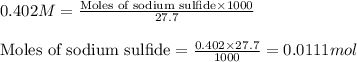
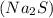 produces 2 moles of sodium ions
produces 2 moles of sodium ions 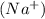 and 1 mole of sulfide ions
and 1 mole of sulfide ions