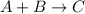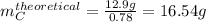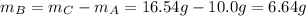Chemistry, 30.03.2020 20:11 sydneyfarrimonp9k3j4
Compound A reacts with Compound B to form only one product, Compound C, and it's known the usual percent yield of C in this reaction is 78%. Suppose 10.0 g of A are reacted with excess Compound B, and 12.9g of Compound C are successfully isolated at the end of the reaction.
1. What was the theoretical yield of C?
2. How much B was consumed by the reaction?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 06:40
Which statement is usually true about the relationship between activation energy and reaction rates? low activation energy barriers result in low rates. high activation energy barriers result in low rates. low activation energy barriers result in no reaction. high activation energy barriers result in no reaction.
Answers: 3

Chemistry, 22.06.2019 10:00
How many grams of co(g) are there in 74.5 ml of the gas at 0.933 atm and 30o c?
Answers: 1

Chemistry, 22.06.2019 17:30
Which scenario is most similar to the type of collision that gas particles have according to kinetic molecular theory
Answers: 1

Chemistry, 22.06.2019 23:00
Arectangle has a diagonal 20 inches long that forms angles of 60 and 30 with the sides. find the perimeter of the rectangle. for geometry
Answers: 3
You know the right answer?
Compound A reacts with Compound B to form only one product, Compound C, and it's known the usual per...
Questions

Mathematics, 03.02.2021 01:00

History, 03.02.2021 01:00


Physics, 03.02.2021 01:00

Social Studies, 03.02.2021 01:00


Mathematics, 03.02.2021 01:00



Social Studies, 03.02.2021 01:00

Mathematics, 03.02.2021 01:00


Mathematics, 03.02.2021 01:00

Mathematics, 03.02.2021 01:00

Mathematics, 03.02.2021 01:00

English, 03.02.2021 01:00


Mathematics, 03.02.2021 01:00