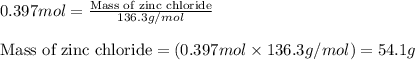Chemistry, 30.03.2020 20:29 liamgreene90
How many grams of ZnCl2 will be produced from 26.0g of Zn and 42.0g of HCl. Round to three significant figures

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 04:30
Which is a chemical property of iron? a. it forms iron oxide (rust) when exposed to moisture and air. b. it is a gray–black metal that is hard to the touch. c. it has a melting point of 2795°f (1536°c). d. it is a good conductor of heat
Answers: 2


Chemistry, 22.06.2019 20:00
Phosphoric acid is a triprotic acid ( =6.9×10−3 , =6.2×10−8 , and =4.8×10−13 ). to find the ph of a buffer composed of h2po−4(aq) and hpo2−4(aq) , which p value should be used in the henderson–hasselbalch equation? p k a1 = 2.16 p k a2 = 7.21 p k a3 = 12.32 calculate the ph of a buffer solution obtained by dissolving 18.0 18.0 g of kh2po4(s) kh 2 po 4 ( s ) and 33.0 33.0 g of na2hpo4(s) na 2 hpo 4 ( s ) in water and then diluting to 1.00 l.
Answers: 3

Chemistry, 22.06.2019 21:30
Liquid ammonia is produced at high temperatures and under great pressure in a tank by passing a mixture of nitrogen gas and hydrogen gas over an iron catalyst. the reaction is represented by the following equation. n2(g) + 3h2(g) → 2nh3(g) changing all but one experimental condition will affect the amount of ammonia produced. that condition is a) increasing the concentration of both reactants b) changing the temperature within the tank c) decreasing the pressure within the tank. d) increasing only the amount of nitrogen present.
Answers: 1
You know the right answer?
How many grams of ZnCl2 will be produced from 26.0g of Zn and 42.0g of HCl. Round to three significa...
Questions

Mathematics, 14.09.2020 21:01

Mathematics, 14.09.2020 21:01

Chemistry, 14.09.2020 21:01

English, 14.09.2020 21:01

Mathematics, 14.09.2020 21:01

Chemistry, 14.09.2020 21:01

Geography, 14.09.2020 21:01

Mathematics, 14.09.2020 21:01

Mathematics, 14.09.2020 21:01

Chemistry, 14.09.2020 21:01

English, 14.09.2020 21:01

Mathematics, 14.09.2020 21:01

Biology, 14.09.2020 21:01

Physics, 14.09.2020 21:01

Arts, 14.09.2020 21:01

Mathematics, 14.09.2020 21:01

Physics, 14.09.2020 21:01

English, 14.09.2020 21:01

Mathematics, 14.09.2020 21:01

Chemistry, 14.09.2020 21:01

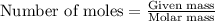 .....(1)
.....(1)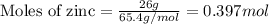
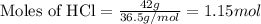
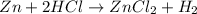
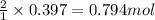 of HCl
of HCl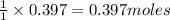 of zinc chloride
of zinc chloride