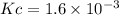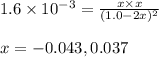Chemistry, 30.03.2020 21:15 4804102262
HI decomposes to H2 and I2 by the following equation: 2HI(g) → H2(g) + I2(g);Kc = 1.6 × 10−3 at 25∘C If 1.0 M HI is placed into a closed container and the reaction is allowed to reach equilibrium at 25∘C, what is the equilibrium concentration of H2 (g)?

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 22:30
Which process describes vaporization that takes place below the surface of a liquid? condensation melting boiling evaporation
Answers: 1

Chemistry, 23.06.2019 01:30
How does the attraction between particles affect the ability of a solvent to dissolve in a substance
Answers: 1

Chemistry, 23.06.2019 06:30
Aplanet similar to earth has four moons roughly the same distance away. the moon that will most affect tides on the planet is the one that has the greatest a) mass. b) volume. c) density. d) amount of water.
Answers: 1
You know the right answer?
HI decomposes to H2 and I2 by the following equation: 2HI(g) → H2(g) + I2(g);Kc = 1.6 × 10−3 at 25∘C...
Questions








Mathematics, 24.03.2021 23:30


Mathematics, 24.03.2021 23:30

Computers and Technology, 24.03.2021 23:30

Mathematics, 24.03.2021 23:30

Mathematics, 24.03.2021 23:30

English, 24.03.2021 23:30


Arts, 24.03.2021 23:30


Health, 24.03.2021 23:30

Mathematics, 24.03.2021 23:30

Mathematics, 24.03.2021 23:30

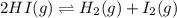
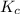 for above equation follows:
for above equation follows:![K_c=\frac{[H_2][I_2]}{[HI]^2}](/tpl/images/0571/2457/ef85e.png)