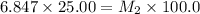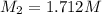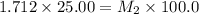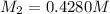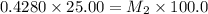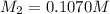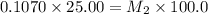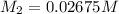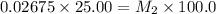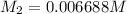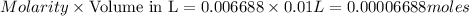Chemistry, 30.03.2020 21:08 studentms5313
Starting with a 6.847 M stock solution of HNO3, five standard solutions are prepared via serial dilution. At each stage, 25.00 mL of solution are diluted to 100.00 mL. Determine the concentration of and the number of moles of HNO3 in the final (most dilute, Md5) solution.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 03:00
Explain how the integumentary system plays a crucial role in the ability to maintain homeoestasis
Answers: 1

Chemistry, 22.06.2019 14:30
1) describe the physical layout of the ocean floor ? 2) explain how the dumbo octopus swims differently than other octopus species and why this would be an advantage in the aphonic zone . 3) why are the types of organisms that live at each underwater hot vent so dramatically different ?
Answers: 3

Chemistry, 22.06.2019 21:30
How many liters of 3.0 m naoh solution will react with 0.60 liters of 4.0 m h2so4? h2so4 + naoh → na2so4 + h2o 1.2 l 1.6 l 2.4 l 2.8 l
Answers: 3

You know the right answer?
Starting with a 6.847 M stock solution of HNO3, five standard solutions are prepared via serial dilu...
Questions






Mathematics, 15.12.2019 14:31


Biology, 15.12.2019 14:31




Mathematics, 15.12.2019 14:31

Mathematics, 15.12.2019 14:31

Mathematics, 15.12.2019 14:31



Mathematics, 15.12.2019 14:31



Mathematics, 15.12.2019 14:31

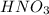 in the final solution is 0.006688 M and number of moles are 0.00006688
in the final solution is 0.006688 M and number of moles are 0.00006688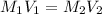
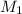 = molarity of stock solution = 6.847 M
= molarity of stock solution = 6.847 M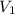 = volume of stock solution = 25.00 ml
= volume of stock solution = 25.00 ml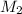 = molarity of ist dilute solution = ?
= molarity of ist dilute solution = ?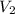 = volume of first dilute solution = 100.0 ml
= volume of first dilute solution = 100.0 ml